swissdamed registration fee
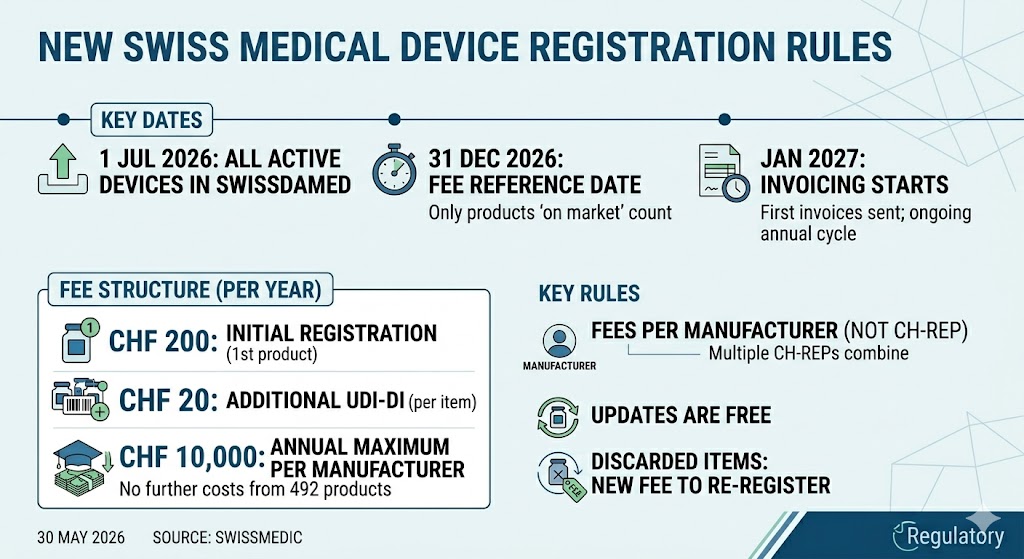
New Registration Requirement for Medical Devices: What Applies from July 2026 As of July 1, 2026, medical devices on the Swiss market must be registered in the national database swissdamed.…

New Registration Requirement for Medical Devices: What Applies from July 2026 As of July 1, 2026, medical devices on the Swiss market must be registered in the national database swissdamed.…

Webinar: Global UDI 2026 -EUDAMED, swissdamed & TGA Australia Deadlines, Transition Periods and Practical Upload Solutions for Small and Large Companies UDI requirements are becoming mandatory in several important markets…
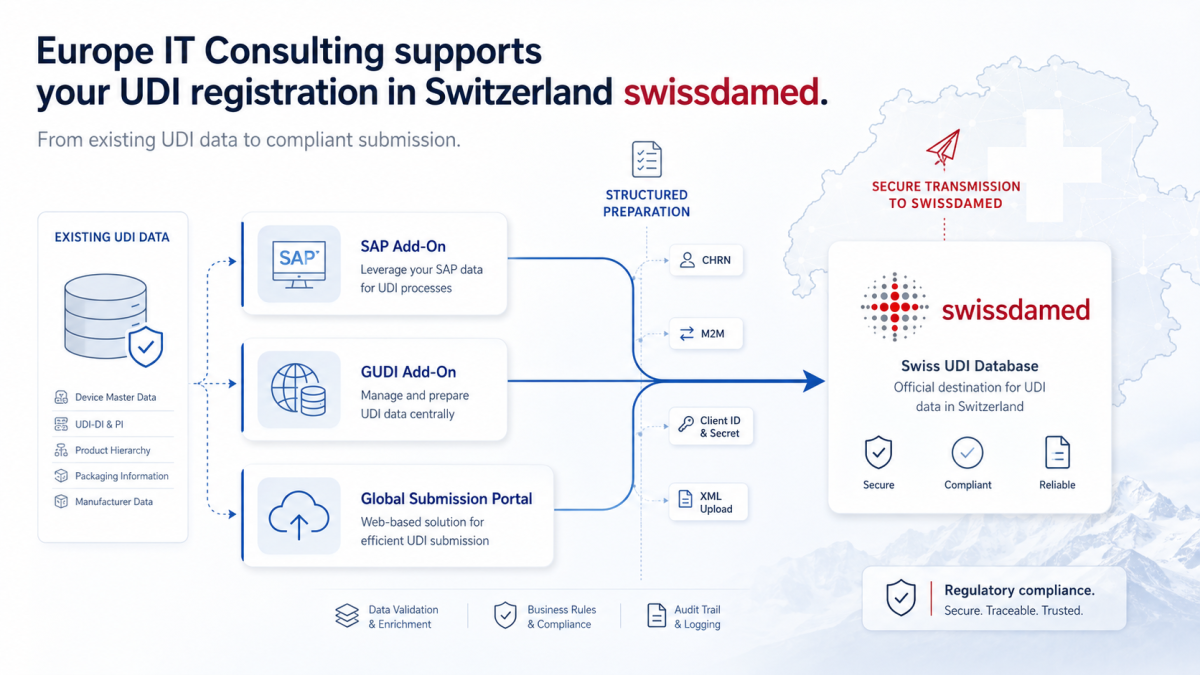
swissdamed: How Europe IT Consulting Supports UDI Registration in Switzerland With swissdamed, Swissmedic is systematically expanding the national database for medical devices and in vitro diagnostics. For manufacturers, Swiss Authorised…

EUDAMED deadline 28 May 2026: When a new UDI-DI brings forward the registration of the Basic UDI-DI In just a few weeks, on 28 May 2026, the use of the…
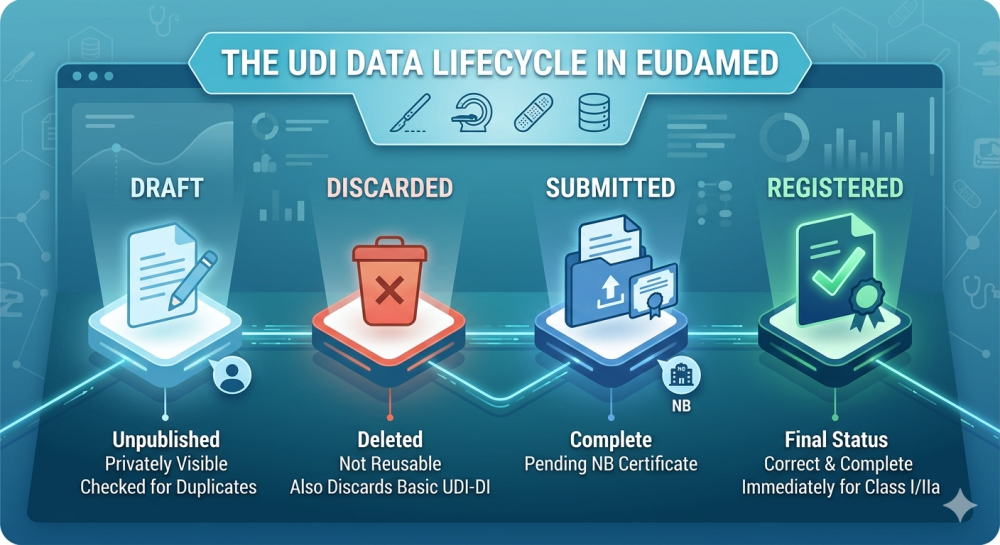
EUDAMED Status What do Draft, Discarded, Submitted, and Registered mean for UDI data? Anyone maintaining UDI data in EUDAMED or transmitting it via XML/M2M will regularly encounter different dataset statuses.…
2026 increases the pressure to act for manufacturers 2026 will be operationally demanding for many MedTech companies. In the EU, the first four EUDAMED modules will become mandatory from 28…
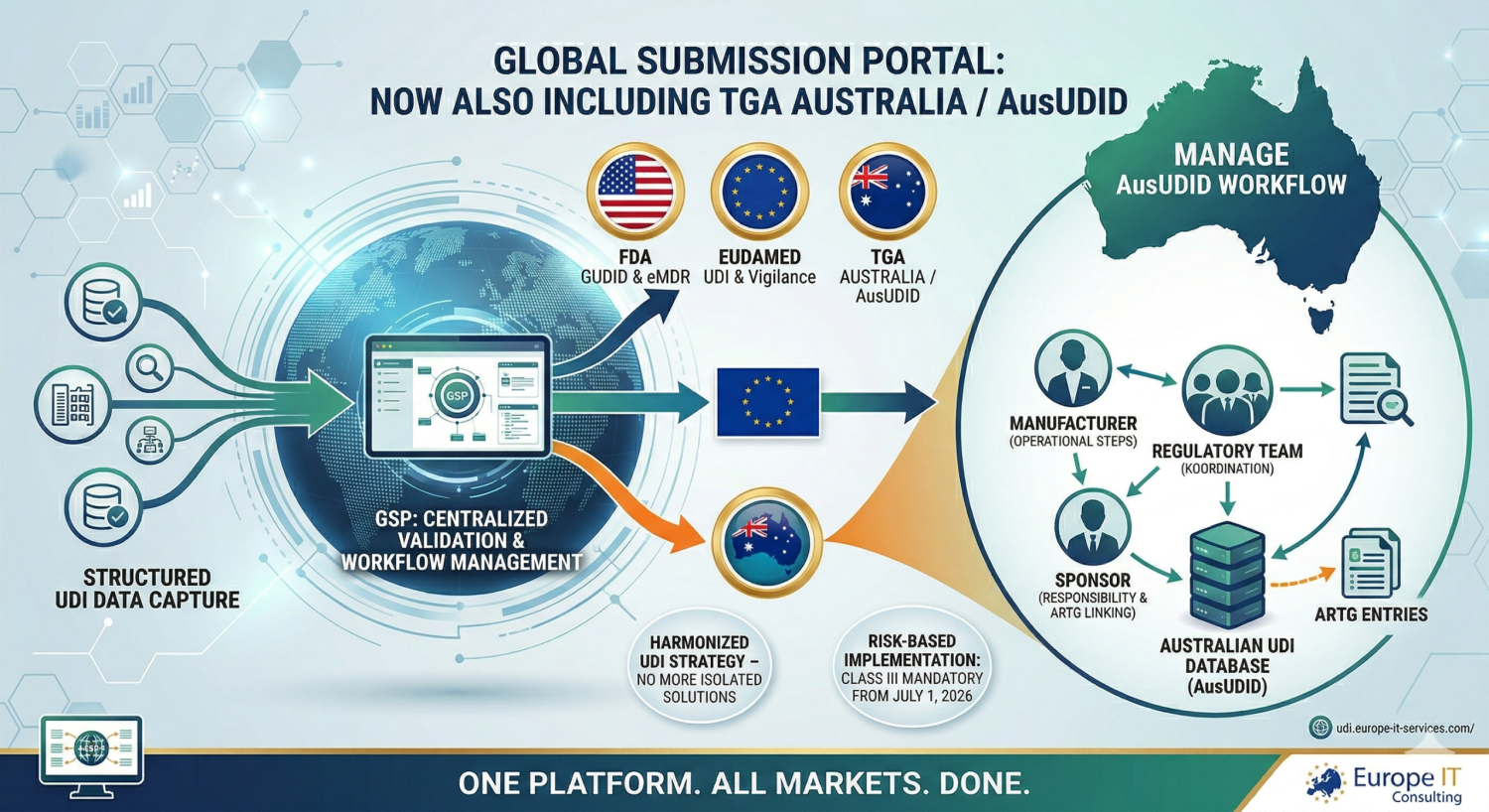
Australia in the GSP: Capture UDI data in a structured way, validate it, and centrally manage it for the AusUDID workflow The Global Submission Portal continues to grow: following its…
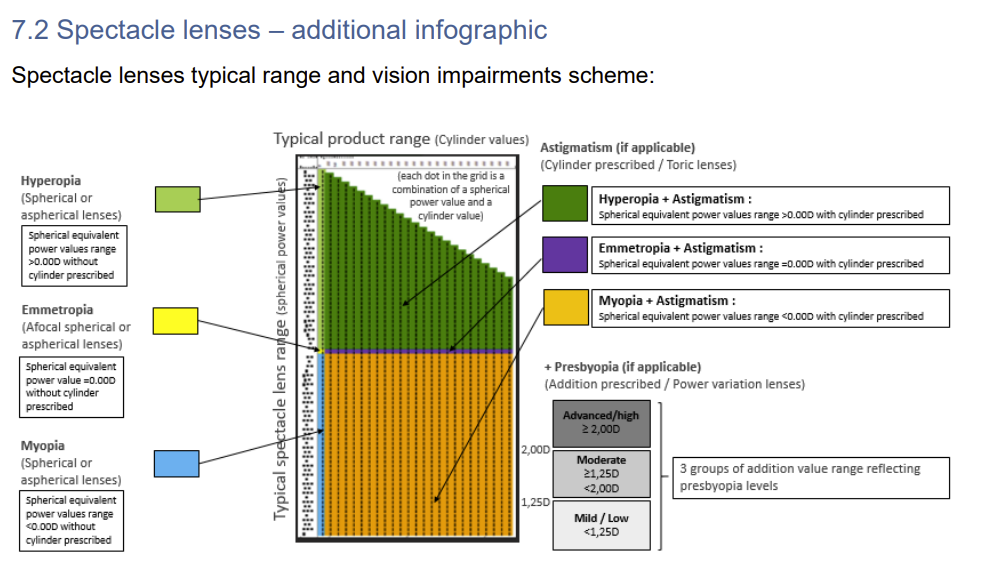
New MDCG Guidance on the Master UDI-DI for spectacle frames, spectacle lenses and ready-to-wear reading spectacles published With the document MDCG 2025-8 Rev. 1, the Medical Device Coordination Group published…

regularia 2026: personal conversations, strong market impulses, and valuable insights for digital regulatory practice Once again, regularia 2026 showed how strongly the medical technology industry is operating in the tension…
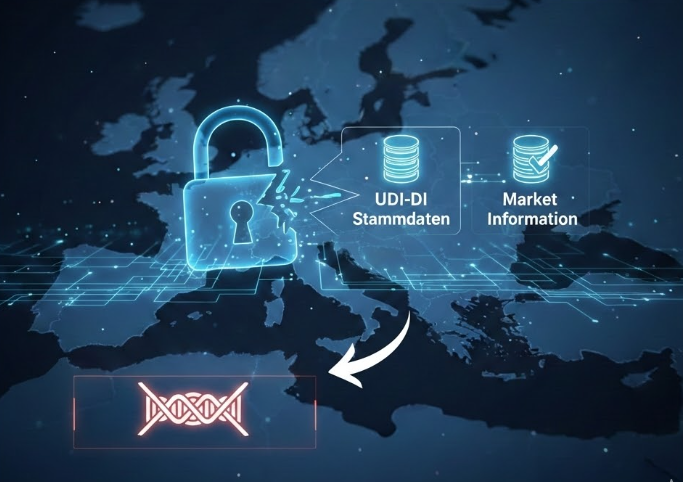
EUDAMED Data Management: Europe IT Consulting GmbH Uncovers Critical Error Sources in UDI Updates The experts at Europe IT Consulting GmbH have identified a significant vulnerability in the EUDAMED architecture…