Global Submission Portal: now TGA Australia / AusUDID as well
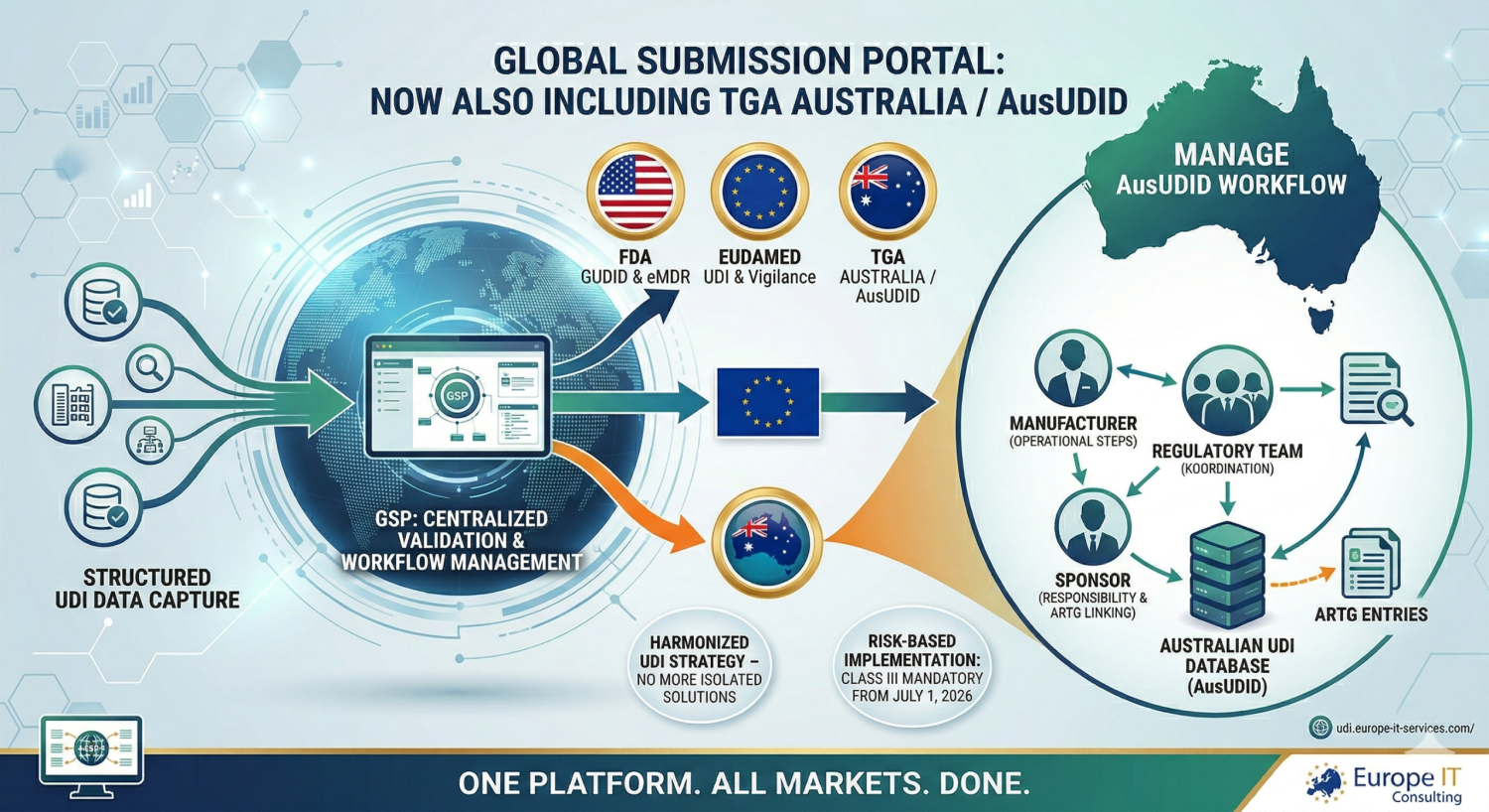
Australia in the GSP: Capture UDI data in a structured way, validate it, and centrally manage it for the AusUDID workflow The Global Submission Portal continues to grow: following its…

Australia in the GSP: Capture UDI data in a structured way, validate it, and centrally manage it for the AusUDID workflow The Global Submission Portal continues to grow: following its…
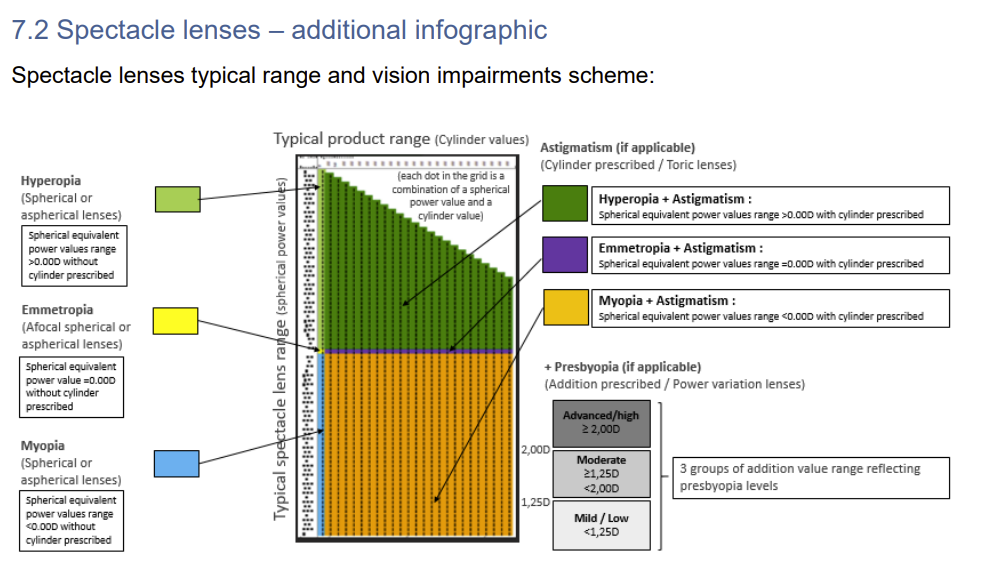
New MDCG Guidance on the Master UDI-DI for spectacle frames, spectacle lenses and ready-to-wear reading spectacles published With the document MDCG 2025-8 Rev. 1, the Medical Device Coordination Group published…

regularia 2026: personal conversations, strong market impulses, and valuable insights for digital regulatory practice Once again, regularia 2026 showed how strongly the medical technology industry is operating in the tension…