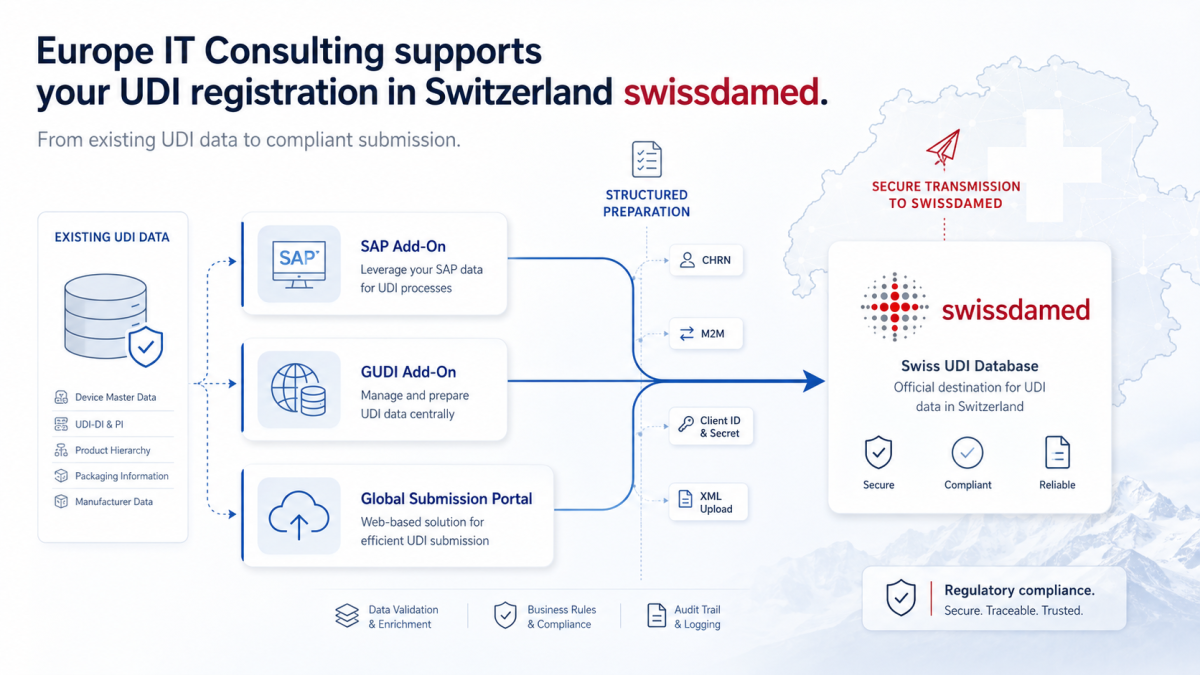
UDI (Unique Device Identification) is a globally standardized system for the identification of medical devices ...................
UE, Suiza, Australia y EE. UU.
Su socio para el cumplimiento global de UDI. Optimizamos y automatizamos la carga de datos en las bases de datos de registro de la UE, Suiza, Australia y EE. UU., garantizando el cumplimiento seguro de todos los requisitos regulatorios.