Global UDI Data Management Solution as SAP Add-On
Visit our Dedicated GUDI Landing Page
Are you looking for a solution specifically designed for managing high data volumes?
On our specialized landing page, we show you how to efficiently scale and validate complex UDI submissions.
One of the challenges in implementing Unique Device Identification is the submission of UDI data to the relevant authorities (such as EUDAMED via AS4, FDA GUDID via AS2/HL7 XML, etc.)
Our UDI SAP Add-On Solution assists you in maintaining your UDI-relevant products and ensures seamless UDI data submission, so you can effortlessly and quickly stay EUDAMED compliant and with all UDI authorities worldwide.
Comprehensive data management for SAP
Our global UDI solution (Unique Device Identification) offers comprehensive data management for SAP (OnPremise and Cloud), supported by a modular structure and Excel integration. With installation on S/4 HANA, SAP Netweaver 7.50 or higher, and the Private S/4 Hana Cloud, you can efficiently manage UDI data.
The frontend based on SAP Fiori and SAP Ui5 enables browser-based access. Additionally, the solution is compatible with the labeling solution from OPAL Labelmanagement, ensuring seamless labeling. Moreover, our solution offers the possibility of data migration and data maintenance using a user-friendly UDI Excel Upload Template, allowing you to quickly upload your UDI data to our add-on.
Are you interested?
Please fill out the form for a quote
Features of our Global UDI SAP Add-On Solution
 Global UDI SAP Add-On |
|
|
| User-friendly SAP Fiori & SAP UI5 | Browser-based easy operation even for non-SAP users |
||
|
Modular structure
For each authority there is a separate plugin, which can be installed as needed.
Available Plugins:
|
||
|
|
||
|
|
||
|
|
||
|
|
||
Watch our global UDI solution video
Our SAP Add-On solution is not only quick and easy to install but also flexible in terms of required UDI authority support and pricing.
Supported by S/4 HANA, SAP Netweaver 7.50 and higher, as well as the Private S/4 Hana Cloud, our solution offers a frontend base on SAP Fiori and SAP Ui5 technology, accessible via web browser.
The Key Benefits of a UDI Software Solution
Experience the benefits of our global UDI solution. As a manufacturer of medical devices, you face the challenge of ensuring the safety and effectiveness of your products while meeting the requirements of regulatory authorities worldwide.
A Global Unique Device Identification solution can help you overcome these challenges while optimizing your business processes.
Are you interested?
Please fill out the form for a quote
Advantages of Dividing UDI Data into Business Units
With “Business Units”, you can assign all UDI data, which are based on your internal materials, to different business units.
What are “Business Units”? These represent your business areas responsible for a group of UDI data. For example, company locations in different countries or different product categories – you decide!
You define the business units at the beginning and give them individual designations. Permissions for which user has access to which data can be easily set by assigning the business units.
Efficient Organization and Access Control
- SAP standard permissions form the basis for assigning permissions. Therefore, nothing changes for your authorization department in terms of organization.
- Business Units allow structured organization of your medical devices by different business units.
- Users receive customized permissions that only give them access to the business units and data relevant to their role.
For efficient organization and easy access control, you can assign all your materials to different business units, simplifying the display and maintenance of certain product groups.
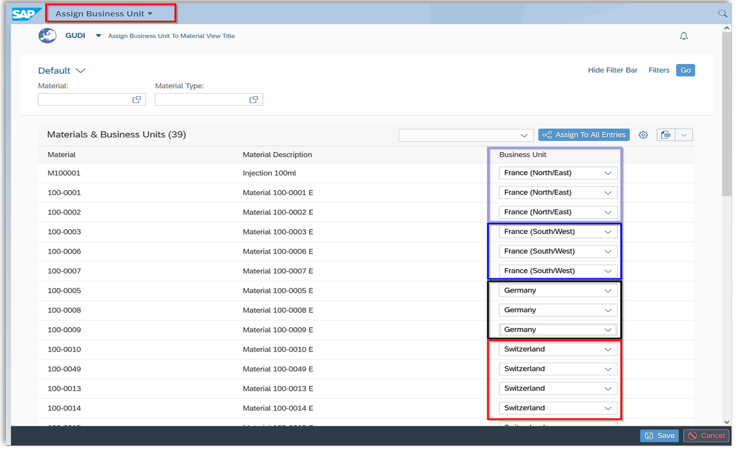
Business Units in our Global SAP Add-On
UDI Approval Process
The approval process for UDI records offers maximum flexibility and adaptability. You decide how many people should be involved in the approval process. You will be notified and visually informed about which data needs to be approved.
Through comprehensive configuration, you can fully customize the approval process to meet your needs and ensure that your UDI records are always accurate and up-to-date.
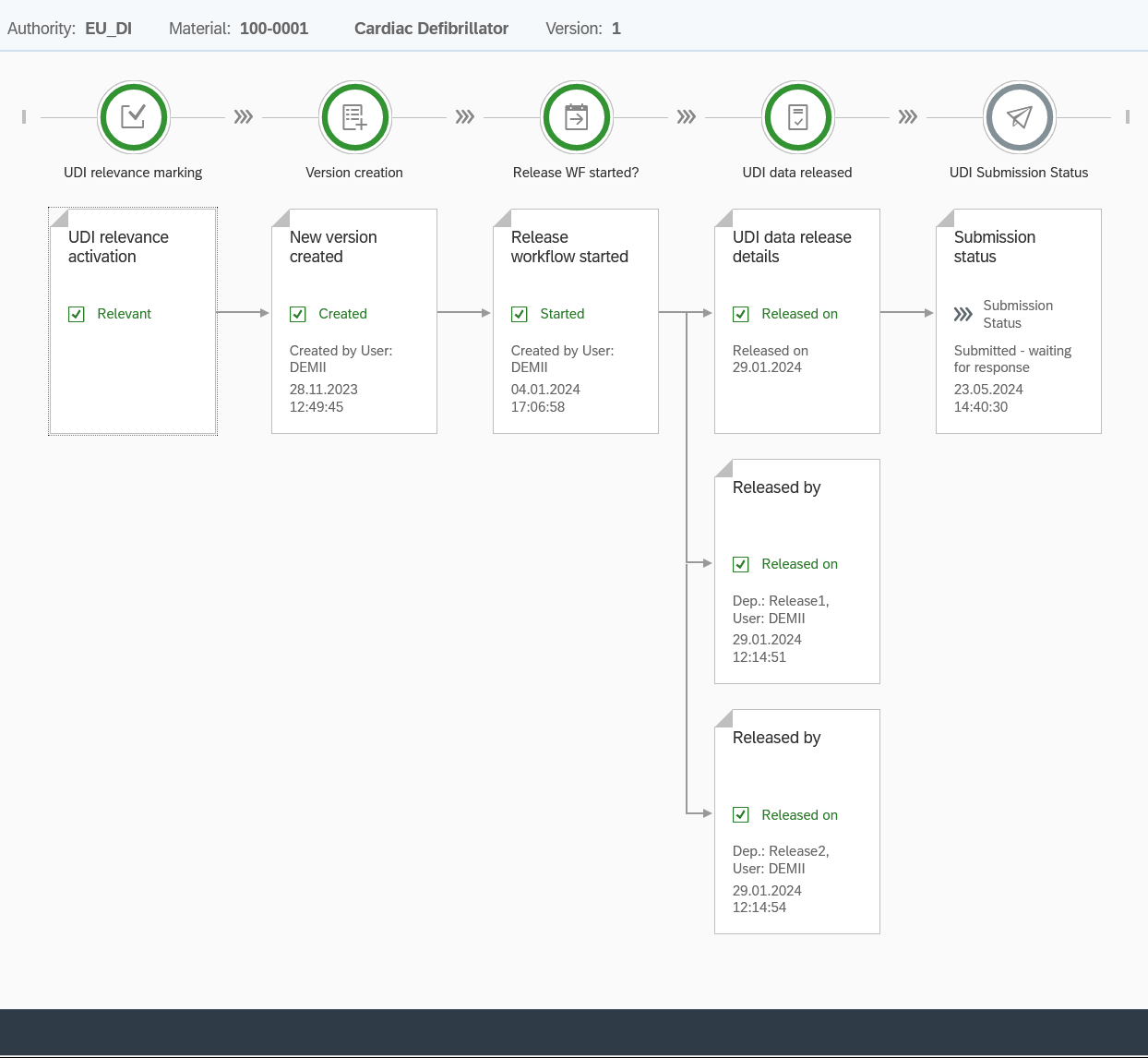
UDI Release Workflow
Features:
- Notification system: Receive real-time notifications via email or visually through the SAP UDI Launchpad Dashboard when approval is required.
- Visual overview: Use our dashboard to quickly and easily see the status of all pending approvals.
- Configurable workflows: Adapt the approval process to your specific requirements, including the number of required approval steps.
- Audit trail: All approval processes are thoroughly documented to ensure complete transparency and traceability.
- User rights: Define user roles and rights to ensure that only authorized persons can grant approvals.
- Automatic reminders: Receive automatic reminders of pending approvals to avoid delays.
- Integration: Our approval process seamlessly integrates into your existing systems and workflows.
- History and traceability: Track all changes and approvals with a detailed history.
Flexibility through available plugins for each UDI authority

UDI SAP Add-On in the SAP Launchpad
We have created Fiori groups for each UDI authority to enable you to manage your products according to the characteristics and requirements of the respective regulatory authorities.
Each authority has its own dedicated Fiori group, whether EUDAMED, FDA, SFDA, etc.
The UDI data manager can thus be assigned dedicated UDI authorities, so they can only see and manage the data they are responsible for.
Depending on which UDI plugins you have installed, only these Fiori groups will be displayed in your Fiori dashboard.
Existing UDI data in the country-specific plugins is reused, minimizing data maintenance effort.
Seamless integration into the Fiori Launchpad makes the user experience intuitive and efficient, allowing you to easily manage your compliance and data management processes.
Are you interested?
Please fill out the form for a quote
Workflow-driven approval process
In regulated industries like medical device manufacturing (Medtech Industry), it is necessary to have an audit trail not only to be successfully validated by an auditor but also to track any type of data change.
In our Global UDI Solution, we have added an audit trail function where you can find all changes to UDI records.
Additionally, we have created a workflow overview. This allows you to see all important information throughout the UDI process of a UDI record. Starting with details on when and by whom the record was marked as UDI relevant, to information on approval and data submission.
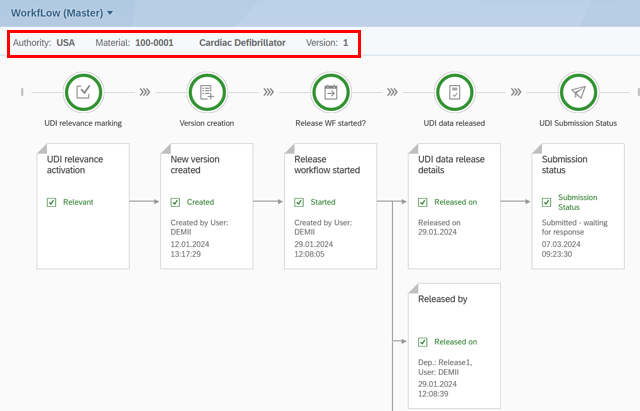
UDI Release Workflow
Import/export UDI data in Excel and easy data migration
Efficient management and transfer of Unique Device Identification (UDI) data are crucial for meeting regulatory requirements. With our comprehensive approach to data migration and the export of UDI data as Excel or XML files, we offer you a reliable solution to manage and transfer your data safely and accurately.

UDI Excel Template
Benefits of UDI Data Export as Excel File
Exporting UDI data in Excel formats offers numerous benefits:
- User-friendliness: Excel files are easy to create and edit.
- Flexibility: Data can be easily analyzed and further processed.
- Compatibility: Excel files are widely used and supported by many applications.
Benefits of UDI Data Export as XML Files
Exporting UDI data in XML formats offers several advantages:
- Structured data: XML files allow for a clear and structured representation of UDI data.
- Automation: XML files can be easily processed and integrated by other systems.
- Standardization: XML is a widely used format accepted by many regulatory authorities.
Data migration and updating UDI data with an Excel template
Uploading UDI data from Excel files is an essential part of data migration. Here are the steps to effectively upload your UDI data:
- Preparation of the Excel file: Fill in your UDI data in the provided Excel template.
- Data validation: Check the Excel file for completeness and accuracy of the UDI data.
- Data upload: Upload the Excel file to our system. Our software validates the data and seamlessly integrates it into your existing UDI database.
Are you interested?
Please fill out the form for a quote
OPAL Label Management and Global UDI Data Management
A complex process on the way to compliance is ensuring that labels meet the requirements of various authorities and country requirements. To facilitate this process, OPAL and Europe IT Consulting have adapted their solutions so that the labeling system can now access all data in the UDI Add-On. Thus, already recorded properties of the medical devices can be printed on the UDI label without detours
The OPAL Label Management is an integrated label management system connected to SAP and incorporated into the Europe IT Consulting solution. It supports the design, printing, and management of labels, including compliance and barcode labels. It also offers label verification features to ensure regulatory compliance.
Learn more about:
MDR Whitepaper – everything you need to know about UDI compliant labeling
Project Implementation and Execution
Our Global UDI Solution supports you in the efficient implementation and execution of your projects. With thoughtful tools and pre-configured templates, your UDI project will proceed in a structured and smooth manner.
Standard Project Template
For smooth project implementation, we offer you standard project templates. These templates are specifically designed to meet the requirements of UDI implementation and provide you with a clear structure for your project. Each template contains detailed instructions and steps to guide you through the entire process.
Pre-configured CSV and Validation Documents
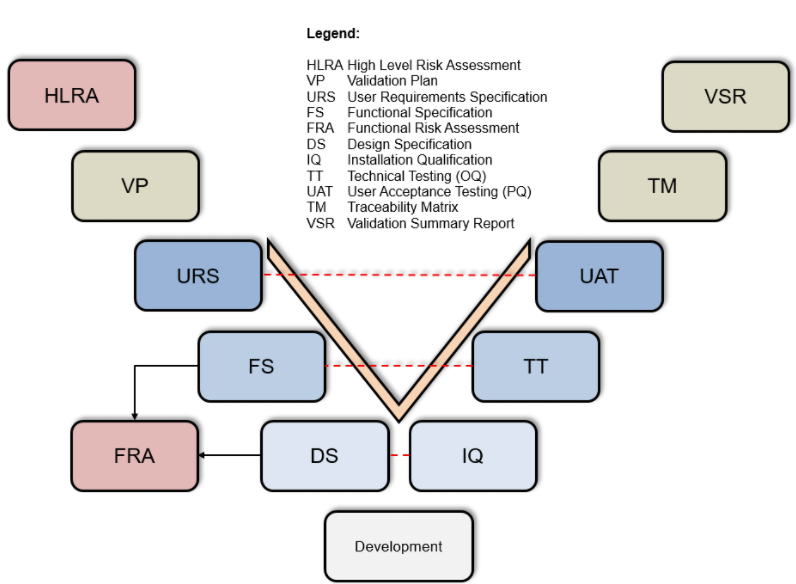
V-Model CSV Validation
To support your data management processes, we offer pre-configured CSV and validation documents. These templates facilitate the capture and validation of your UDI data to ensure all regulatory requirements are met. The CSV templates are designed to capture your data consistently and completely, while the validation documents help you verify data integrity.
By using our standard project templates and pre-configured CSV and validation documents, the implementation of your UDI solution will be more efficient and targeted. These tools are designed to minimize project effort and ensure compliance so you can focus on the essential aspects of your business.
Discover how our comprehensive project support tools can help you successfully implement your UDI projects and meet all regulatory requirements.
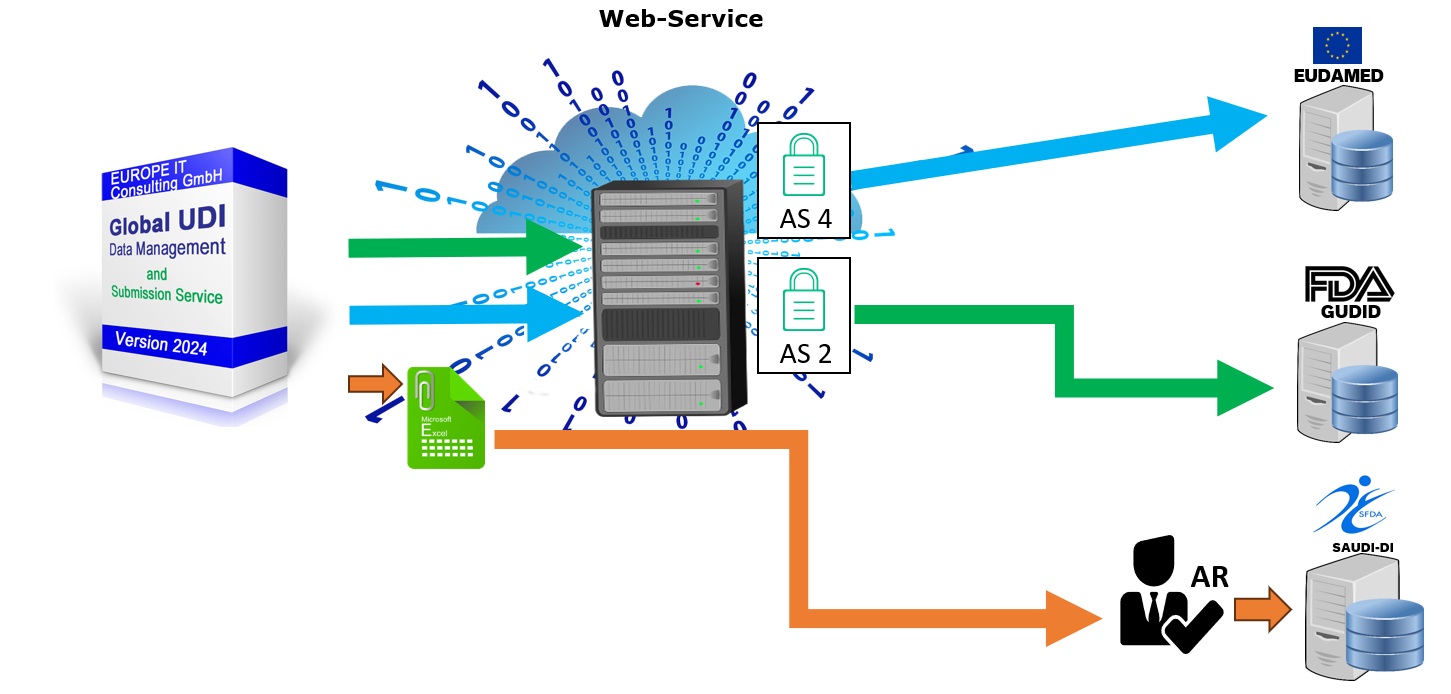
UDI AS2/AS4 Data submission
Our Global UDI Solution provides comprehensive support for the transfer of your UDI data to the respective regulatory authorities. We ensure that your data is transferred correctly and completely to ensure compliance with global requirements.
Seamless Data Transmission
With our Global UDI Solution, you can transfer UDI data seamlessly and efficiently. The solution supports submission to various authorities such as EUDAMED, FDA, SFDA, and others. Integration with existing systems and the use of pre-configured templates significantly simplifies data transmission.
Ensuring Data Integrity
Our solution provides comprehensive validation mechanisms to ensure that all UDI data is correct and complete before transmission. This minimizes the risk of errors and ensures that your data meets regulatory requirements.
Automated Workflows
Using automated workflows, you can optimize the transmission process. These workflows include the creation, validation, approval, and transmission of your UDI data. Each step is thoroughly documented to ensure full traceability.
Customer Success Stories
Our software and services have proven themselves multiple times and are already successfully used by many customers from the medical industry.
Since the introduction of FDA UDI requirements in 2016, numerous companies have successfully used our SAP add-on to manage their UDI data independently, integrate it into SAP, and transfer it to the relevant databases.
Our UDI data management solution for medical device manufacturers enables seamless integration and efficient management of UDI data, leading to significant process improvement. See for yourself through the voices of our customers…
 |
|
|
|
Are you interested?
Please fill out the form for a quote
UDI Data Transfer Without SAP? Yes,..
UDI data must be prepared and compiled for upload to the databases of the relevant authorities. The easiest way to do this is to enter all the information into a template, the UDI Excel Template from Europe IT Consulting GmbH, with multiple tabs.
Our UDI Excel Templates are designed to provide an easy and efficient way to manage your UDI data, even if you do not use an SAP system. These templates cover all the necessary fields and information required to meet UDI requirements.
Since we regularly adapt these templates to changes and new requirements, you can be sure of always being compliant and interacting with the UDI databases (EUDAMED, FDA, etc.) in the best possible way. Our UDI Excel template ensures that your data is captured correctly and completely, making the transfer to the relevant authorities seamless.
With the UDI Excel Templates from Europe IT Consulting GmbH, you can:
- Capture UDI data easily and structured: All necessary information is organized in clear tabs.
- Ensure your data is compliant: The templates are regularly updated to meet the latest regulatory requirements.
- Interact efficiently with the UDI databases: Our templates are designed to facilitate the upload process to databases like EUDAMED and FDA.
By using our UDI Excel Templates, companies without SAP systems can effectively manage their UDI data and ensure they meet all regulatory requirements.
Nehmen Sie Kontakt mit uns auf
UDI News and Articles
Current State of Worldwide UDI Systems
The UDI (Unique Device Identification) field continues to evolve and is more or less advanced depending on the country.
If you are in any way involved in the business of medical devices, it may be relevant for you to stay updated on the regulations and deadlines worldwide.
Webinars on the Topic of Unique Device Identification
Here you will find a list of our upcoming online events, as well as a selection of past webinars, recordings, and related materials available to you.
Our webinars are specifically designed to provide you with in-depth knowledge and practical insights into the world of UDI. Whether you are new to this field or have already gained experience and want to deepen your knowledge, our webinars offer something for everyone.
Your Contacts
Our dedicated team consists of highly qualified consultants and experts with extensive experience in the UDI industry. With a passion for technology and innovation, we are committed to providing our clients with the best solutions and first-class service.

Ugur Müldür

Ismail Demiralp
Are you interested?
Please fill out the form for a quote
Keywords: UDI, CSV, GAMP5, SAP, UDI Software, UDI Excel Template, AS2, AS4, XML, Data Transfer, UDI Manager, MDR, IVDR, IVDD, EUDAMED, FDA, SFDA, NMPA, TGA, GTIN, UDI-DI, Basic UDI-DI, AIMDD, GS1 Datamatix, EMDN, SRN, CHRN
