Global Submission Portal: The New Era of Regulatory Compliance

Unified platform for global FDA and EUDAMED submissions now available
Europe IT Consulting is pleased to announce the release of the Global Submission Portal – a revolutionary solution designed to simplify and accelerate regulatory compliance processes for medical device manufacturers worldwide.
The Challenge
Medical device manufacturers face the complex task of registering and reporting products to various authorities globally. Each authority has its own requirements, formats, and processes, leading to:
- Time-consuming manual processes
- High risk of error during data entry
- Lack of transparency regarding submission status
- Fragmented workflows across multiple systems
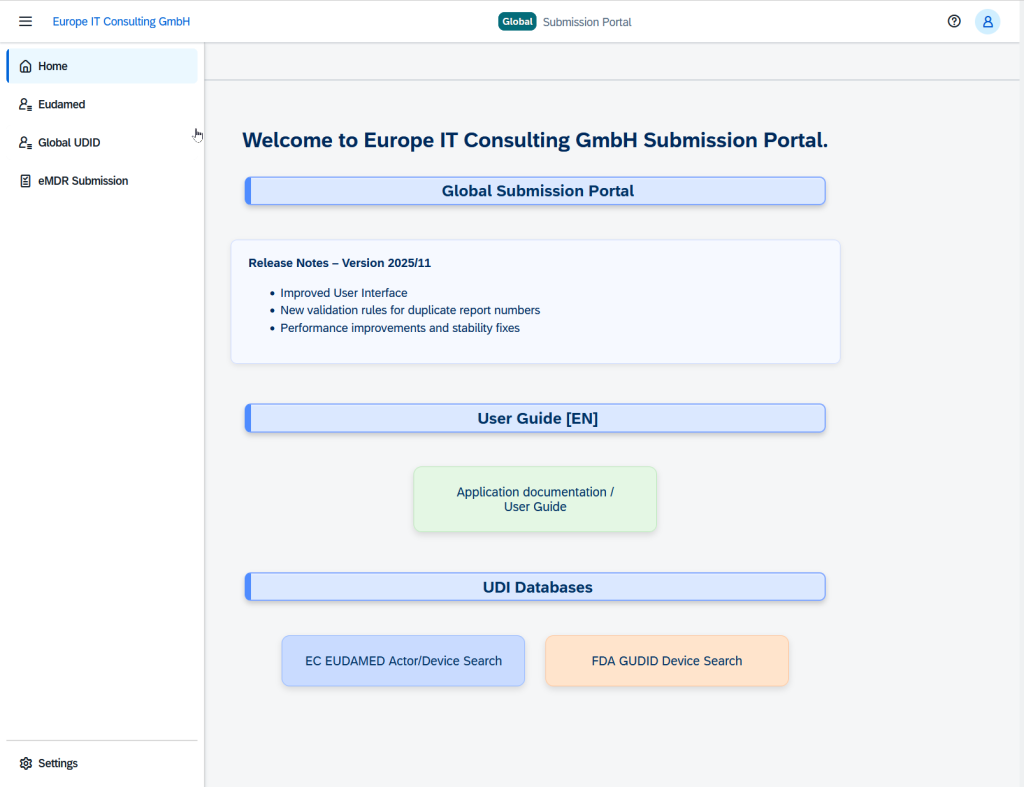
The Solution: Global Submission Portal
Our Global Submission Portal integrates several submission workflows into a single, user-friendly platform. Manufacturers and regulatory teams can now submit compliance-relevant datasets directly to authorities worldwide – efficiently, securely, and transparently.
Visit our dedicated GSP Landing Page
Are you looking for a solution specifically designed to handle high data volumes? On our specialized landing page, we show you how to efficiently scale and validate complex UDI submissions.
Core Functionality
The portal automates the entire submission process: from Excel-based data collection to automatic validation and direct transmission to the authorities – all in one seamless workflow.
Supported Modules
The portal currently supports four critical compliance areas:
- 🇺🇸 FDA eMDR: Electronic Medical Device Reporting for fast FDA notifications.
- 🇺🇸 FDA GUDID: Global UDI Database for US market registration.
- 🇪🇺 EUDAMED UDI: EU MDR-compliant UDI submissions.
- 🇪🇺 EUDAMED Vigilance: EU Manufacturer Incident Reports.
- AusUDID UDI: Australa UDI
- swissdamed UDI: Switzerland UDI
Key Features in Detail
Excel-Based Upload & High Data Volume
Simply upload your prepared Excel files. Our solution is explicitly designed for high volume, allowing thousands of records to be processed in the shortest possible time. The system automatically recognizes the data structure and prepares the submission.
GUDI – The SAP UDI Add-on for Global Compliance.
Compliance Deadlines: EUDAMED & swissdamed
Regulatory requirements are tightening: the mandatory use of EUDAMED for product registration is approaching. In Switzerland, the registration obligation via swissdamed is also already mandatory for many actors. Our portal helps you meet these deadlines safely through automated processes.
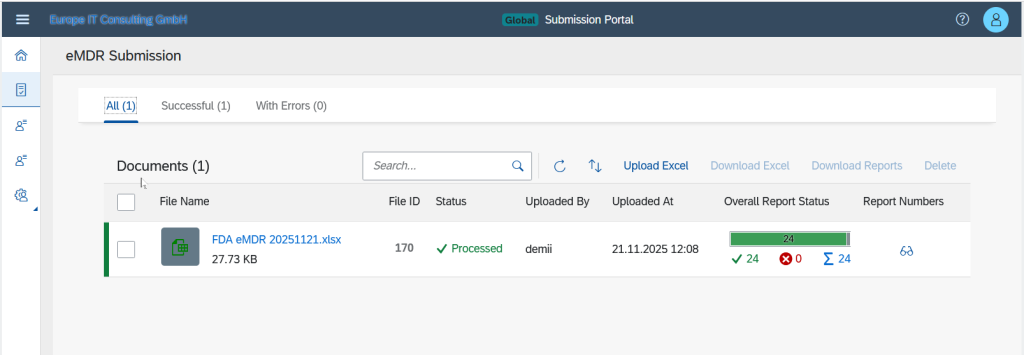
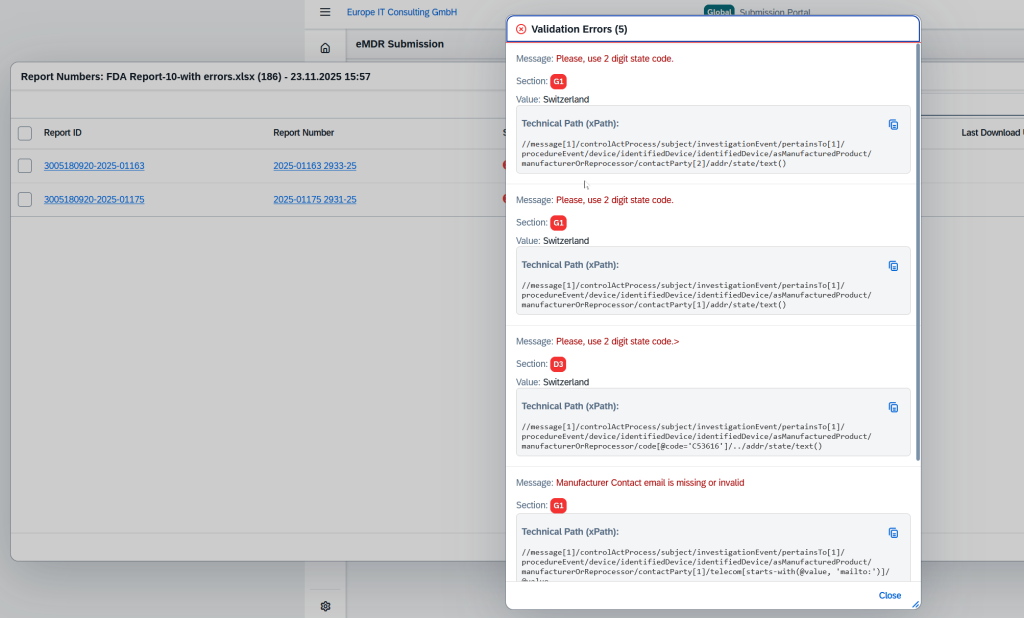
Validation Errors & Real-Time Status
In the event of failed transmissions, detailed validation errors are displayed. Each submission passes through transparent status phases: Uploaded, Processing, Success, or Failed.
Security and Compliance
- Encrypted data transmission (TLS/SSL)
- Strict data isolation per customer
- Audit trail for all activities
Continuous Development
The Global Submission Portal is continuously updated to support new regulatory requirements. Planned expansions include additional international authorities and extra compliance modules.
Shall we meet at Regularia 2026?
Visit us on March 3, 2026, in Tuttlingen. We would be happy to show you our GSP and SAP solutions live on-site!
Conclusion
With the Global Submission Portal, Europe IT Consulting offers a modern, efficient solution for the challenges of global regulatory compliance. Experience for yourself how the portal revolutionizes your workflows!