
regularia 2026: personal conversations, strong market impulses, and valuable insights for digital regulatory practice
Once again, regularia 2026 showed how strongly the medical technology industry is operating in the tension between regulatory requirements, digitalization, and operational feasibility. For Europe IT Consulting, the event was an excellent opportunity not only to take away current developments from presentations and discussions, but above all to put them into perspective through direct exchange with experts, companies, and partners.
The focus was not only on traditional regulatory topics, but also on very specific practical questions:
- How can regulatory requirements be integrated efficiently into existing processes?
- How can the effort involved in UDI, data quality, and data transmission be reduced?
- And how can technical solutions be designed in such a way that they are not only convincing in theory, but also truly work in everyday practice?
What regularia 2026 was about

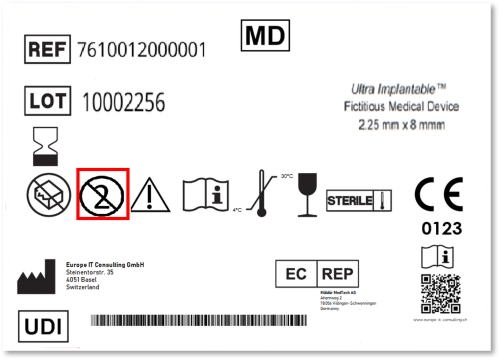
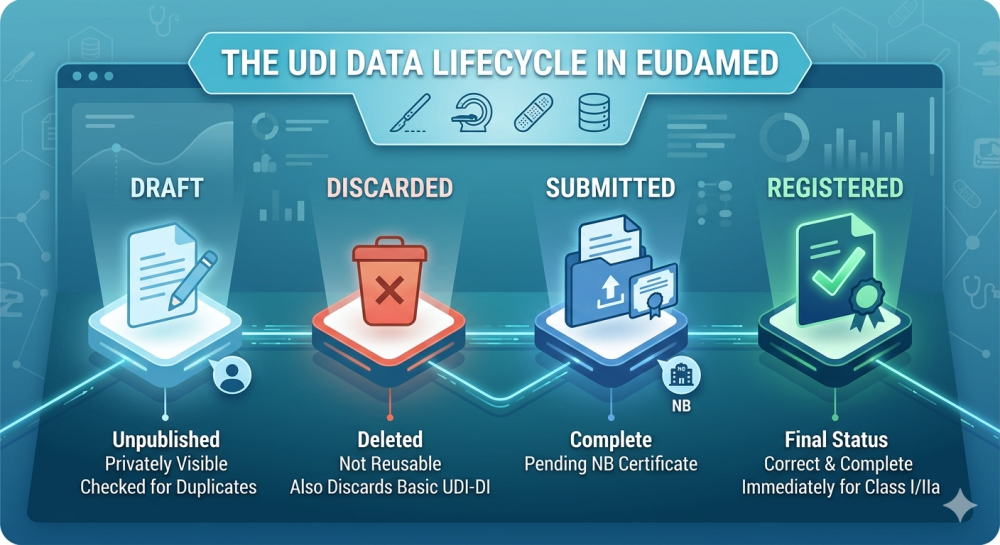
For years, regularia has been an important meeting point for companies, professionals, and partners in the medical technology sector. In 2026, it once again became clear that topics such as MDR, IVDR, UDI, international requirements, data quality, and digital processes can no longer be viewed in isolation. The regulatory landscape continues to evolve, while at the same time many companies are under increasing pressure to make their processes more efficient, transparent, and resilient.

Especially where regulatory requirements meet operational reality, key questions arise:
- How is data maintained in a structured way?
- How can manual work steps be reduced?
- How can different functional areas such as Regulatory Affairs, Quality Management, and IT collaborate more effectively?
These were precisely the topics that shaped many of the conversations and impressions on site.
Our impressions and key conversations

What was particularly valuable for us was how open and concrete many of the conversations were. In numerous discussions, it became clear that many companies continue to face similar challenges: inconsistent data sources, high manual effort, evolved process landscapes, and increasing requirements for traceability and data quality.

At the same time, it was clearly noticeable that the industry is continuing to evolve. Many companies are working intensively to make regulatory processes more digital and more robust. This is no longer just about individual submissions or data records, but about reliable end-to-end processes: from data maintenance and validation to documentation and the actual transmission itself.

The exchange with other companies and partners was also very valuable for us. Events such as regularia create exactly the kind of space where not only current challenges can be discussed, but also new collaborations and shared perspectives can emerge. For us, this is a key added value of such formats.
Why personal presence matters for an IT company

Especially for an IT company, it is important not only to talk about requirements, but to experience them through direct exchange with the market. Many challenges only become truly tangible on site: What questions are companies asking right now? Where do friction points arise in practice? Which solutions are actually needed – and which sound good but fall short in everyday operations?
Personal conversations provide exactly these insights. They help align technical solutions not with theoretical ideal processes, but with real workflows, responsibilities, and regulatory expectations. For us, this is a decisive factor when it comes to supporting companies meaningfully in the areas of UDI, data management, and regulatory data transmission.
regularia 2026 once again confirmed for us how important this closeness to practice is. Anyone who develops or supports digital solutions for regulatory processes must understand the actual challenges companies face – technically, professionally, and organizationally.
Conclusion

We look back on a very valuable regularia 2026. The event delivered many relevant impulses, enabled important conversations, and once again demonstrated how great the need is for practical, integrated, and efficient solutions in the regulatory environment.

We would like to thank everyone we had the opportunity to speak with on site, and we look forward to continuing the exchange.








Related Posts