2026 increases the pressure to act for manufacturers
2026 will be operationally demanding for many MedTech companies. In the EU, the first four EUDAMED modules will become mandatory from 28 May 2026, namely Actor Registration, UDI/Device Registration, Notified Bodies & Certificates, and Market Surveillance. In Switzerland, the registration of products, systems, and procedure packs in swissdamed will become mandatory from 1 July 2026. And in Australia, the mandatory UDI requirements for Class III and Class IIb medical devices will also begin on 1 July 2026.
This increases the pressure not just in one market, but simultaneously across several regulatory systems. For manufacturers, this means that UDI data must not only exist, but must also be available in a form that can be validated, assigned, and submitted in a structured and timely manner. Companies that continue to work with isolated individual processes or manually maintained stand-alone solutions significantly increase the risk of delays, rework, and unnecessary coordination effort. This conclusion follows from the nearly simultaneous compliance dates in the EU, Switzerland, and Australia.
Australia 
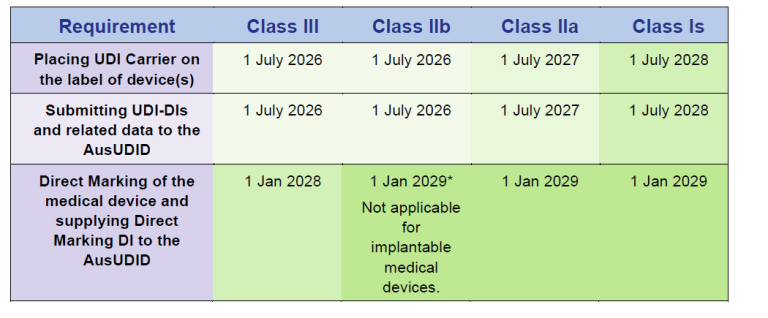
The pressure to act is particularly tangible in Australia. The TGA requires not only UDI labelling, but also, among other things, the submission of UDI data to the AusUDID, the linkage to the ARTG ID, the ongoing maintenance of current datasets, and – for implantable devices – the inclusion of the UDI on the Patient Implant Card. In addition, UDI data must generally be submitted within 30 days after the next supply in Australia. Sponsors may appoint a third party for this purpose, but they remain responsible for the submitted data.
Switzerland 
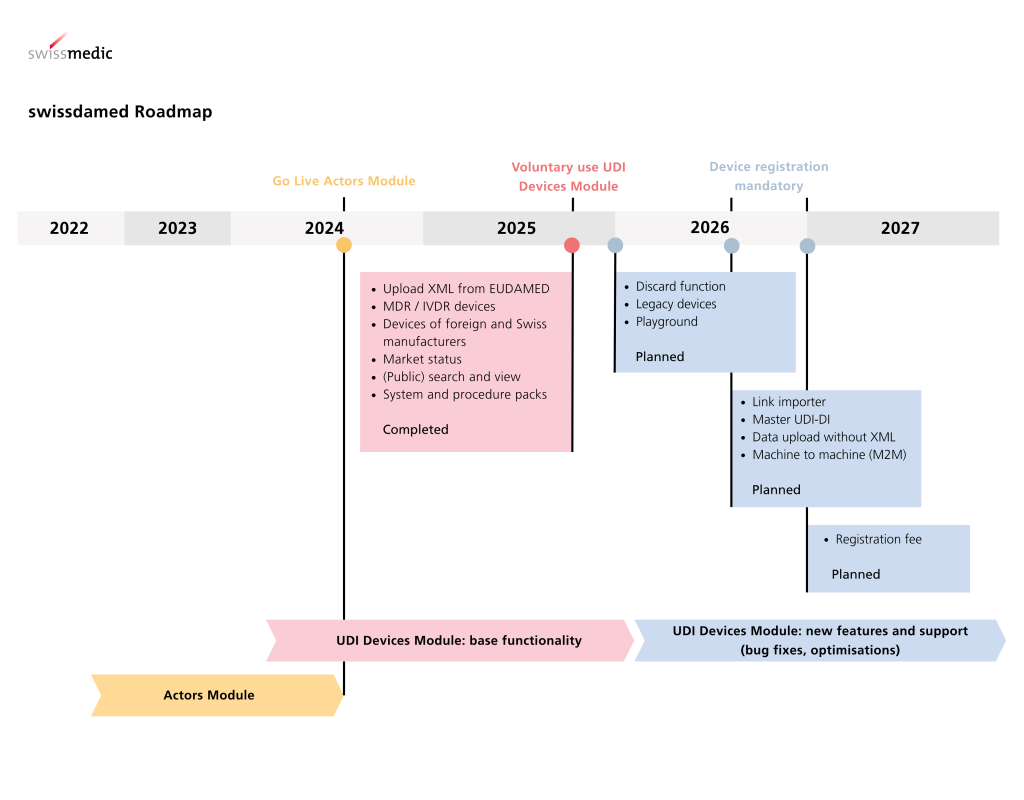
The direction is also clear in Switzerland. Swissmedic names 1 July 2026 as the start date for mandatory product registration in swissdamed and generally grants a transition period until 31 December 2026. At the same time, no transition period applies to certain products, namely where serious incidents, field safety corrective actions, or trends must be reported to Swissmedic. Companies should therefore not rely solely on the calendar year 2026, but should establish robust data and registration processes much earlier.
European Union 
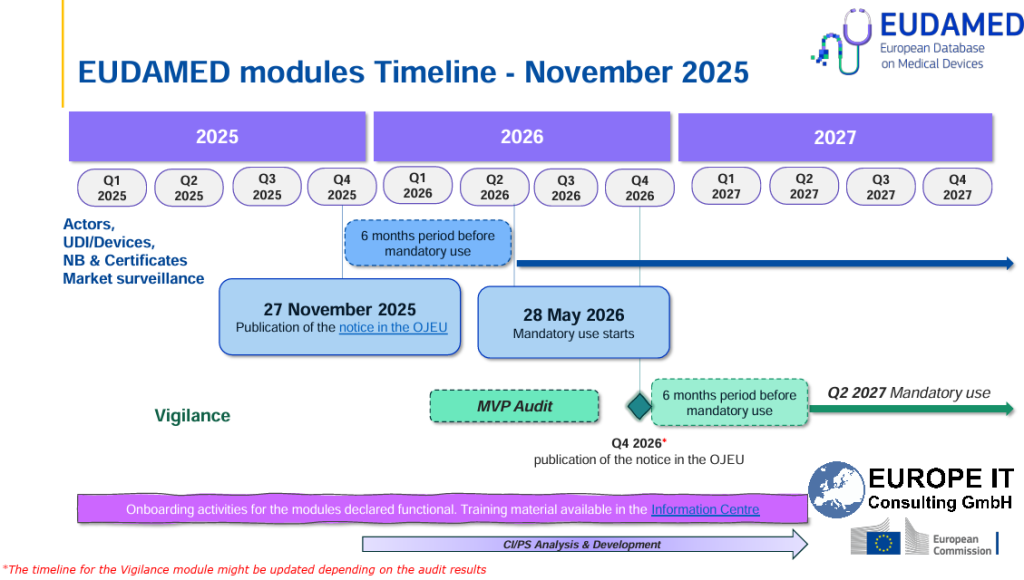
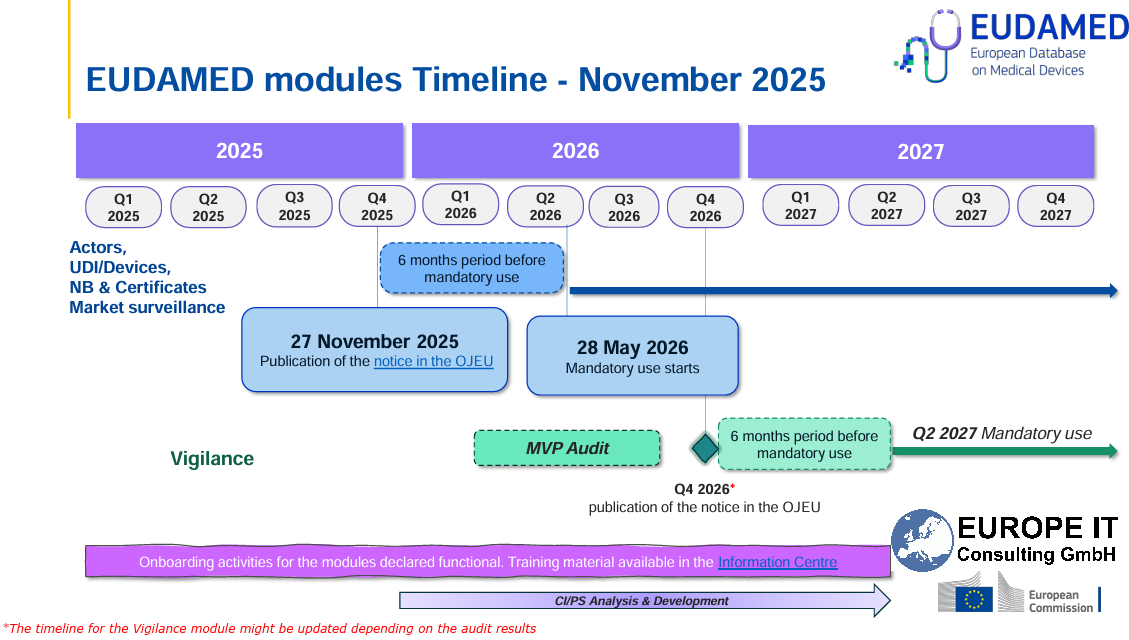
For the EU, this also means that EUDAMED is finally moving from voluntary to mandatory operation. Following the publication of the functionality notice at the end of November 2025, the six-month transition period is running, so that the first four modules will become mandatory from 28 May 2026. In this context, Swissmedic additionally points out that a transition period until 27 November 2026 applies to the corresponding EU registrations. This further increases the pressure on companies to manage their product data ոչ միայն locally or on a project-specific basis, but in a market-ready and submission-ready manner.
Global Submission Portal
This is exactly where our Global Submission Portal comes in. Europe IT Consulting initially introduced the portal for FDA eMDR, FDA GUDID, and EUDAMED UDI, then expanded it to include EUDAMED Vigilance (MIR), and most recently added the workflow for Swissmedic / swissdamed and TGA Australia / AusUDID. The benefit is clear: capture regulatory data in a structured way, validate it, and centrally manage it for multiple authority workflows instead of building new stand-alone processes for each market.
Our conclusion is therefore clear: 2026 is not a year for late reactions. Anyone who needs to keep EUDAMED, swissdamed, and Australia in view at the same time should now harmonize data quality, responsibilities, and submission processes. The earlier this foundation is in place, the lower the friction losses, correction loops, and operational bottlenecks during implementation. The deadlines are set. Now execution matters.
Would you like to prepare your UDI data for EUDAMED, swissdamed, and Australia in a structured way and bring it together in one consistent submission process? Talk to us about the right approach for your products, markets, and data sources.







Related Posts