
The live webinar has already ended, but you are very welcome to register to receive the recording and the presentation materials!
The EUDAMED database plays a central role in MDR and IVDR compliance. But what does this mean for manufacturers, importers, and distributors of medical devices? Which deadlines are crucial, and how can you submit your data efficiently and in compliance with regulations?
In our free expert webinar, we provide a comprehensive overview of the latest EUDAMED developments and show you how to best prepare for upcoming requirements.
What to Expect:
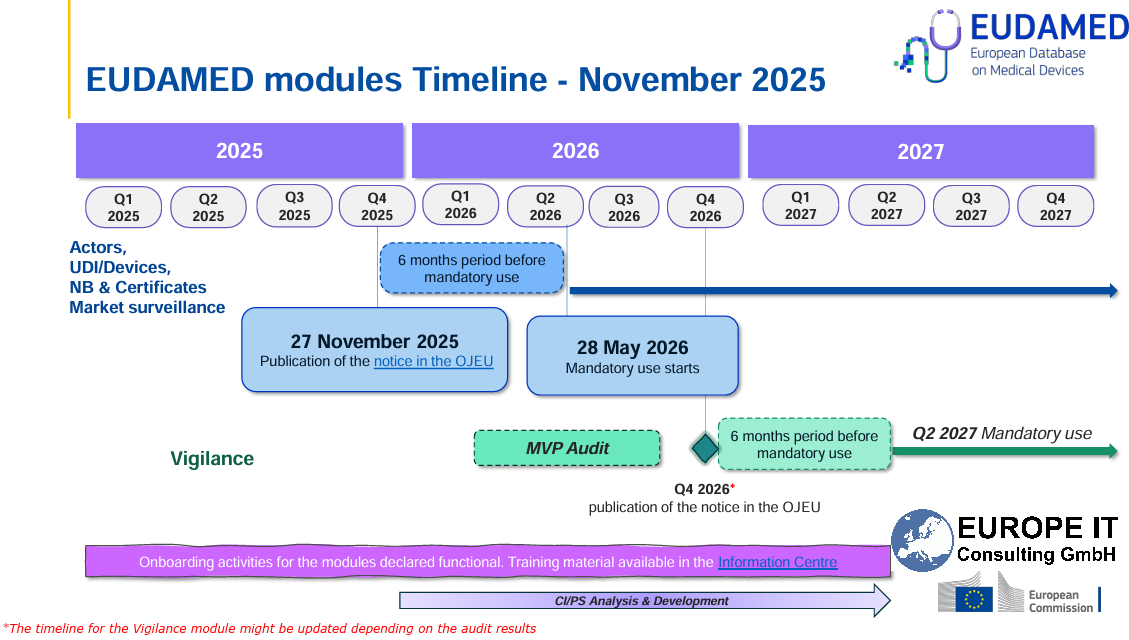
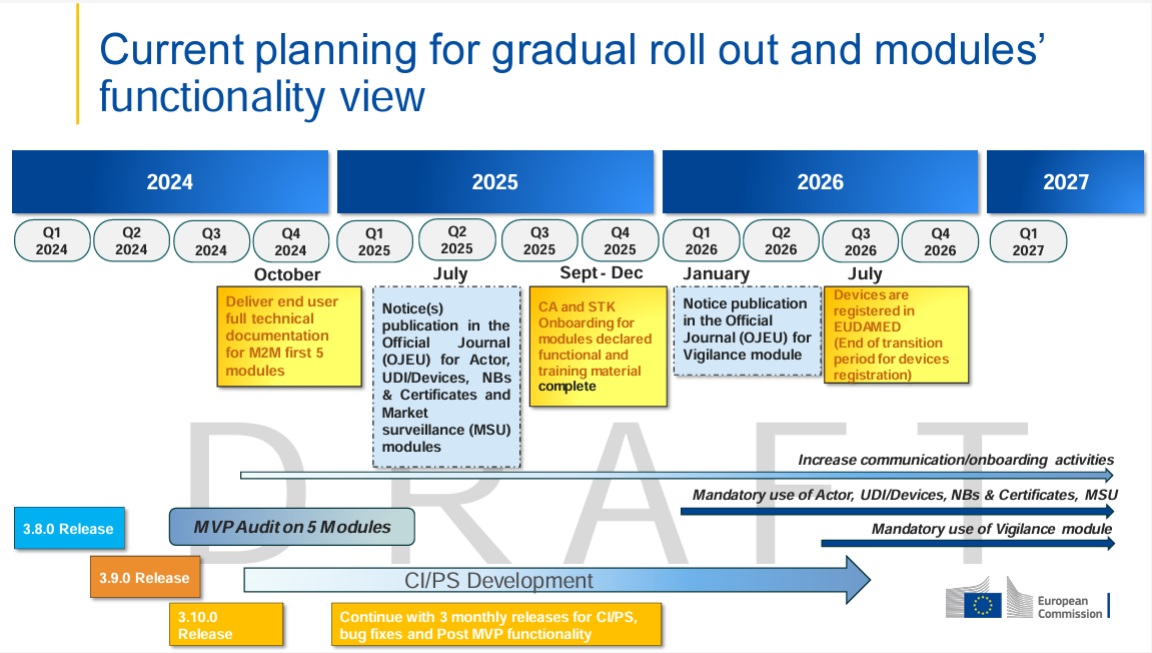
✅ Status & Timeline – Which EUDAMED modules are already active? What deadlines must be met?
✅ EMDN Codes, SRN & Terminology – A clear explanation of key concepts and requirements
✅ How to use the EUDAMED Website – A practical guide to navigating the platform
✅ Data Submission to EUDAMED – Exploring options for efficient and secure data upload
Your Benefits:
🔹 Concise expert knowledge – clear & practical
🔹 Valuable insights for strategic compliance planning
🔹 Live Q&A session with our EUDAMED experts
🚀 The live webinar has already ended, but you are very welcome to register to receive the recording and the presentation materials!








Related Posts