The smart solution for structured AusUDID UDI data management
Prepare your UDI data for the Australian UDI Database in a structured, reviewable and efficient way.
Your challenge is our solution
With the Australian Unique Device Identification Database, AusUDID, the Therapeutic Goods Administration (TGA) is introducing a central UDI database for medical devices and in vitro diagnostics supplied in Australia. For manufacturers, sponsors and other organisations involved, this creates a new operational challenge: UDI data must be prepared, checked and submitted completely, consistently and in line with TGA requirements.
Our AusUDID Excel template provides the first structured step – developed by UDI experts for companies that want to prepare their Australian UDI data efficiently and avoid later validation errors.
Please complete the form to request an offer
The AusUDID UDI Excel template at a glance:
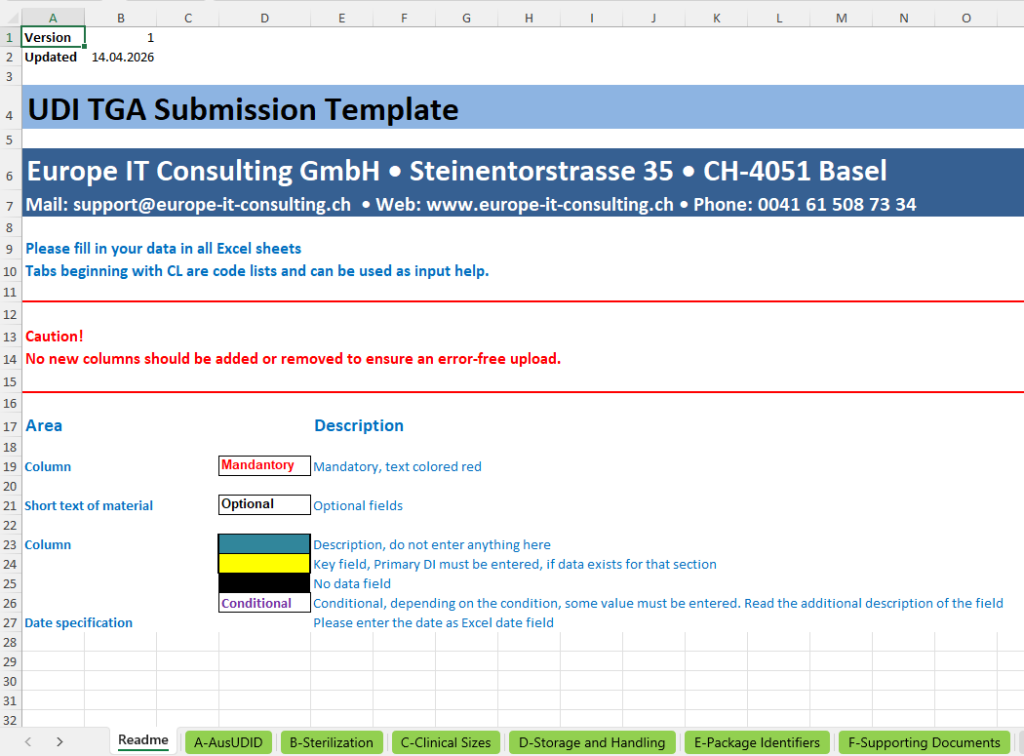
The template is divided into functional data entry tabs and technical code lists.
The actual data entry takes place in tabs A to F. The tabs with the prefix “CL_” contain code lists and selectable values that serve as input support and validation basis. This ensures that data entry is not free and uncontrolled, but based on predefined values and regulatory-relevant selection logic.
A-AusUDID – Central main tab for the UDI data record
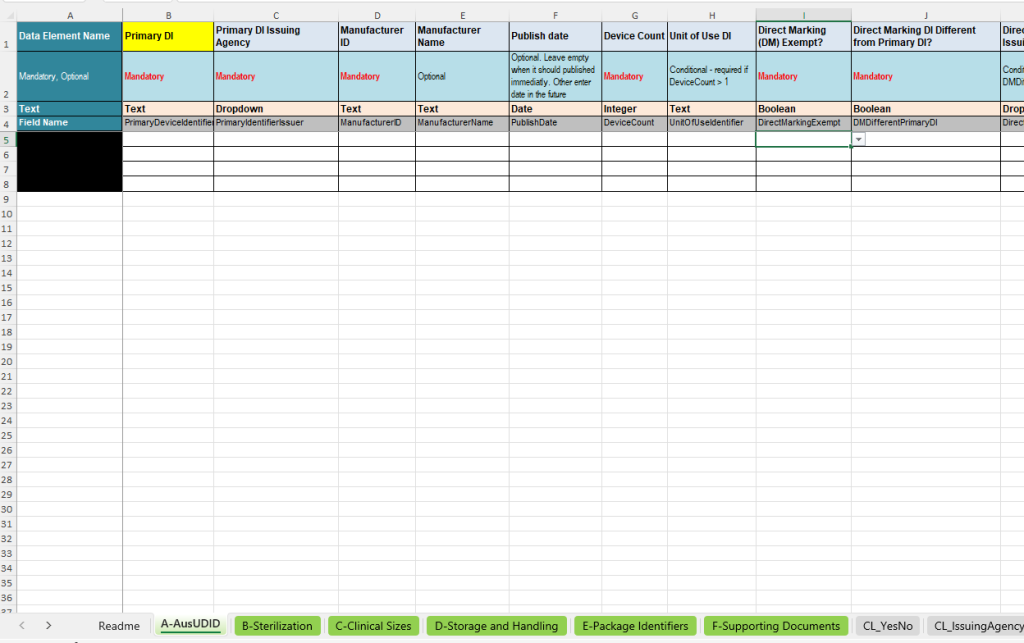
The A-AusUDID tab is the central main tab of the template. This is where the most important master data of a UDI data record is maintained. The Primary Device Identifier forms the central key through which the additional tabs are linked to the respective product.
- Primary Device Identifier: Central device identifier for the unique identification of the product.
- Primary DI Issuing Agency: Specification of the issuing agency, e.g. GS1, HIBCC or ICCBBA.
- Manufacturer ID and Manufacturer Name: Identification and name of the manufacturer.
- Publish Date: Optional publication date if the data record is not to be published immediately.
- Device Count and Unit of Use DI: Recording of the number of included devices and, where required, the Unit of Use identifier.
- Direct Marking: Information on whether direct marking is required, whether an exception applies and whether a different Direct Marking DI is used.
- Brand Name and Model/Version: Product name, model or version designation.
- Device Class: Selection of the Australian risk class, e.g. Class III, Class IIb, Class IIa, Class I or IVD classes.
- Software and kit information: Indication of whether the product is software, Software as a Medical Device or a kit.
- Device Description and GMDN Code: Description of the device and assignment to the Global Medical Device Nomenclature.
- ARTG ID, Catalogue Number and Sponsor Commercial Distribution End Date: Information for linking to the Australian Register of Therapeutic Goods and sponsor-related distribution information.
- MRI Safety Status: Classification of MRI safety, e.g. MR Safe, MR Conditional or MR Unsafe.
- Single Use and Reuse: Information on single use and, where applicable, limited reusability.
- Latex and natural rubber labelling: Recording of relevant labelling information for latex or natural rubber.
- Sterility and sterilisation: Information on whether the device is supplied sterile or must be sterilised before use.
- Production Identifier: Indication of whether lot/batch, manufacturing date, serial number, expiry date or Donation ID Number is present on the label.
- Secondary DI and Previous DI: Documentation of additional or previous device identifiers, including the respective issuing agency.
Benefit: This main tab creates the central data basis for AusUDID submission. It consolidates all essential product, manufacturer, labelling, classification and identification information in one structured table.
Please complete the form to request an offer
B-Sterilization – Sterilisation methods
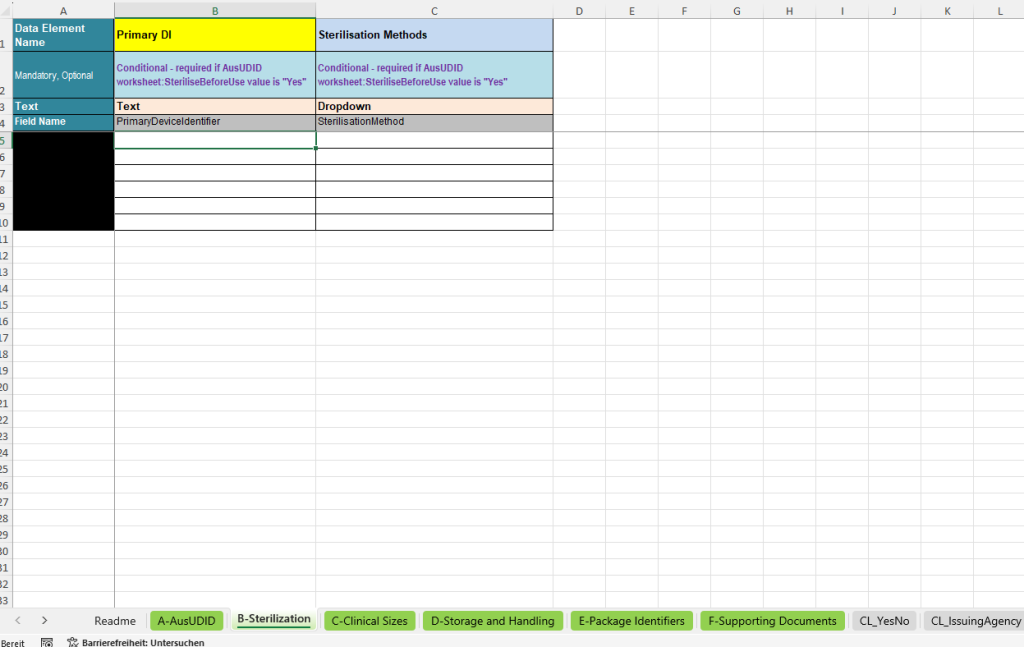
The B-Sterilization tab is used if the main tab indicates that the device must be sterilised before use. In this case, the relevant sterilisation methods must be recorded in a structured way for the respective Primary Device Identifier.
- Primary DI: Link to the corresponding UDI data record from the main tab.
- Sterilisation Methods: Selection of the applicable sterilisation method via a defined selection list.
The template supports different sterilisation methods such as ethylene oxide, steam sterilisation, dry heat, gamma radiation, electron beam, hydrogen peroxide, plasma or other methods.
Benefit: Sterilisation information is not maintained in an unstructured way in the main tab, but managed in a dedicated tab. This allows several relevant methods to be clearly assigned to the respective product.
C-Clinical Sizes – Clinical size information
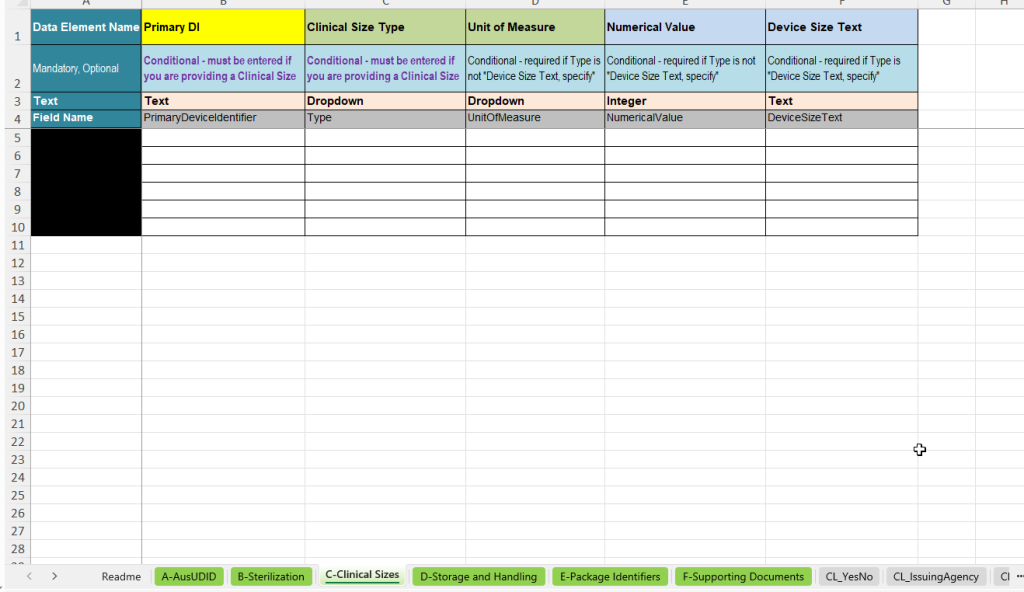
The C-Clinical Sizes tab is used to record clinically relevant size information. It is used when product size, dimension or other relevant size characteristics are to be or must be specified for the AusUDID data submission.
- Primary DI: Link to the corresponding UDI data record.
- Clinical Size Type: Selection of the type of size information, e.g. length, area, angle, gauge or other size categories.
- Unit of Measure: Unit of the size information, e.g. millimetres, centimetres, inches, degrees or other units.
- Numerical Value: Numerical value of the size information.
- Device Size Text: Free-text field if the size cannot be meaningfully represented numerically or should be specified as specific text.
The template distinguishes between numerical size information and pure text-based size information. If a standardised size with unit is provided, type, unit and value are maintained. If the size can only be meaningfully represented as text, the Device Size Text field is used.
Benefit: Clinical sizes are recorded consistently and in a structured manner. At the same time, the template remains flexible enough for products whose size information cannot be represented purely numerically.
D-Storage and Handling – Storage and handling conditions
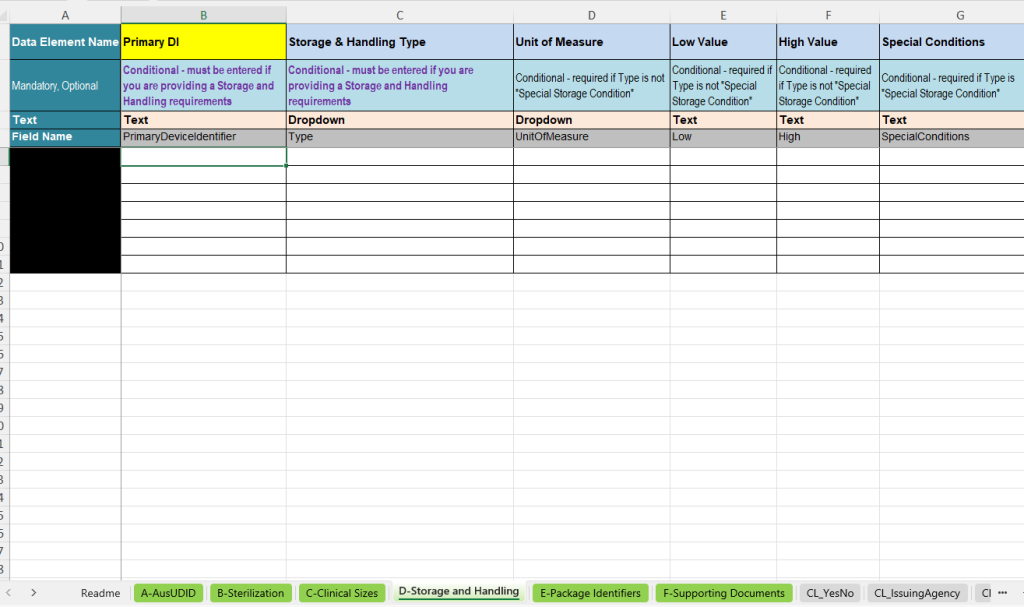
The D-Storage and Handling tab documents special storage and handling requirements for the device. This information is important where certain environmental conditions or specific storage and transport instructions must be observed.
- Primary DI: Assignment of the condition to the respective UDI data record.
- Storage & Handling Type: Selection of the type of condition, e.g. temperature, humidity, atmospheric pressure or special storage condition.
- Unit of Measure: Unit for the respective condition, e.g. degrees Celsius, Fahrenheit, Kelvin, percent relative humidity, kPa or mbar.
- Low Value and High Value: Lower and upper limit value for the corresponding condition.
- Special Conditions: Free-text field for special conditions if no numerical limit values are used.
The template contains logic whereby numerical limit values and a unit are used for measurable conditions. For special storage conditions, a textual description can be maintained instead.
Benefit: Critical storage and handling data are recorded in a structured way and can be reviewed on a product-specific basis. This helps avoid unclear free-text entries and contradictory limit values.
Please complete the form to request an offer
E-Package Identifiers – Package identifiers and packaging hierarchy
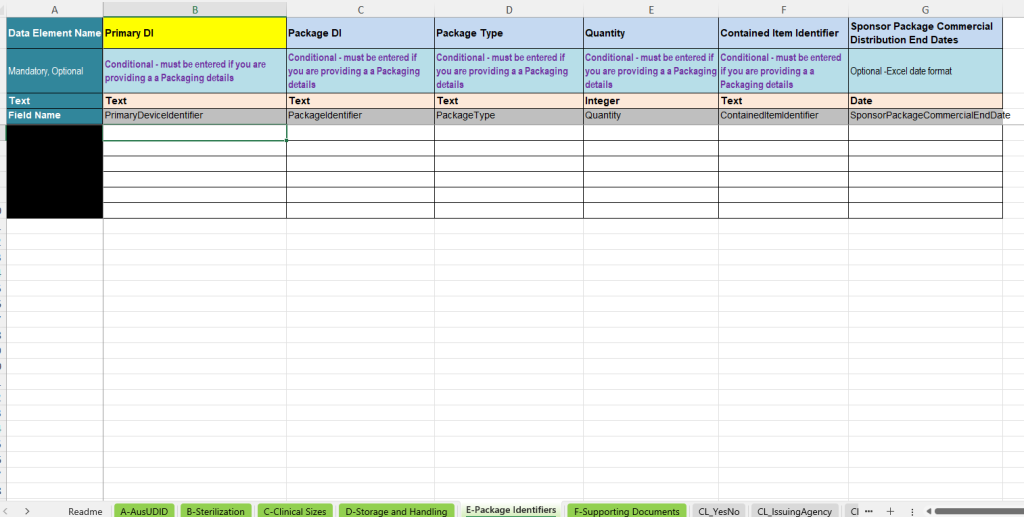
The E-Package Identifiers tab is used to record additional package identifiers and packaging information. It is particularly relevant when further packaging levels or contained units must be documented in addition to the Primary Device Identifier.
- Primary DI: Link to the main data record.
- Package DI: Identifier of the packaging unit.
- Package Type: Type of packaging unit, e.g. packaging level or packaging form.
- Quantity: Number of contained units.
- Contained Item Identifier: Identifier of the contained product or contained unit.
- Sponsor Package Commercial Distribution End Date: Optional end date of commercial distribution for the packaging level.
Benefit: Packaging structures are not merely described as text, but represented as a traceable relationship between package identifier, contained product and quantity. This is particularly important for multi-level packaging, sets or large product portfolios.
F-Supporting Documents – Supporting documents
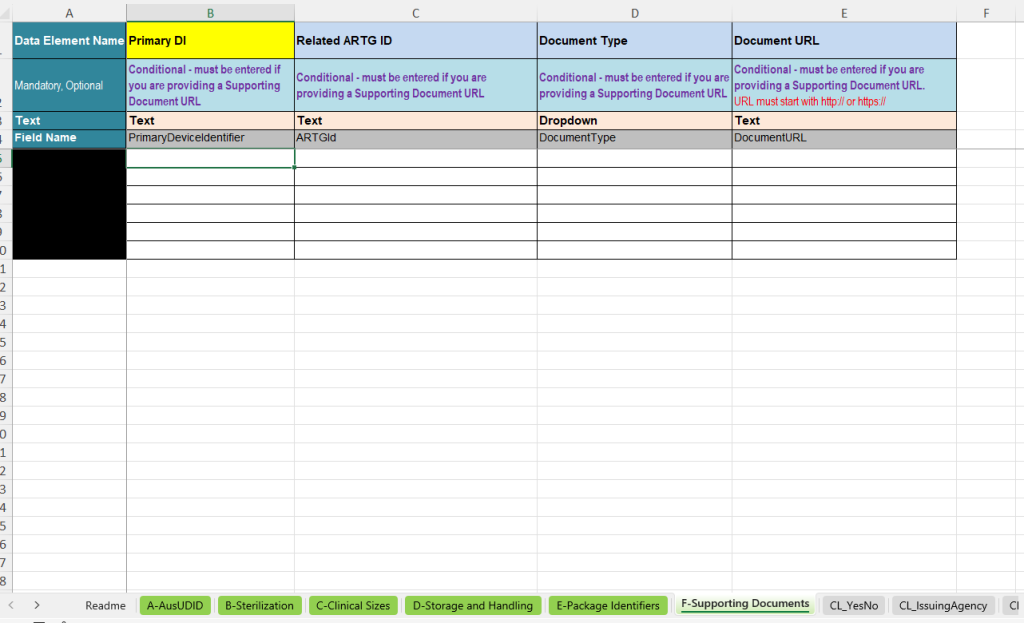
The F-Supporting Documents tab is used when product-related documents are to be specified as URLs. These may include, for example, Instructions for Use or Patient Information Leaflets.
- Primary DI: Link to the corresponding UDI data record.
- Related ARTG ID: Reference to the relevant ARTG ID.
- Document Type: Selection of the document type, e.g. Instructions for Use or Patient Information Leaflet.
- Document URL: Web address of the document. The URL must begin with http:// or https://.
Benefit: Supporting documents are structurally linked with the UDI data record and the ARTG reference. This increases traceability and makes product-related information easier to provide.
Readme – Instructions and editing logic
The Readme tab contains general instructions for using the template. Among other things, it explains that all relevant Excel sheets must be completed and that the tabs with the prefix “CL_” serve as code lists and input support.
- Do not add or remove columns: The structure of the template must not be changed to ensure error-free further processing.
- Mandatory fields: Fields marked in red must be completed.
- Optional fields: Can be completed if the information is available or technically meaningful.
- Conditional mandatory fields: Must be completed if the respective condition is met.
- Date fields: Dates should be maintained as actual Excel date values.
Benefit: The Readme tab reduces user errors and ensures that users understand the logic of the template before they begin data entry.
CL tabs – Code lists and controlled selectable values
The tabs with the prefix CL_ are not normal data entry tabs, but controlled code lists. They serve as the basis for drop-down fields, standardised selectable values and consistent terminology within the template.
- CL_YesNo: Standardised Yes/No and N/A values.
- CL_IssuingAgency: Supported UDI issuing agencies such as GS1, HIBCC and ICCBBA.
- CL_Device Class: Australian risk classes for medical devices and IVDs.
- CL_HandlUnitOfMeasures: Units and types for storage and handling conditions.
- CL_Sterilization: Controlled list of sterilisation methods.
- CL_SupportingDocs: Document types such as Patient Information Leaflet or Instructions for Use.
- CL_DeviceIsSoftware: Selectable values for Software as a Medical Device, medical device with software or “No”.
- CL_MRISafety: MRI safety values, e.g. MR Safe, MR Conditional and MR Unsafe.
- CL_ClinicUnitOfMeasures: Comprehensive unit and type list for clinical size information.
Benefit: The code lists reduce free text, improve data quality and support consistent technical further processing. Users select values from controlled lists instead of entering terms manually and potentially inconsistently.
Please complete the form to request an offer
Intelligent functions for maximum efficiency
- Clear separation of tabs: Master data, sterilisation, clinical sizes, storage conditions, packaging and documents are maintained separately.
- Primary DI as the key: Additional information is clearly linked to the main data record via the Primary Device Identifier.
- Mandatory and conditional logic: The template distinguishes between mandatory fields, optional fields and conditionally required information.
- Drop-down-supported data entry: Code lists support standardised entries and reduce deviations.
- Preparation for validation: The structured template facilitates review before technical preparation or submission.
- Scalability: Suitable for individual products as well as extensive UDI portfolios.
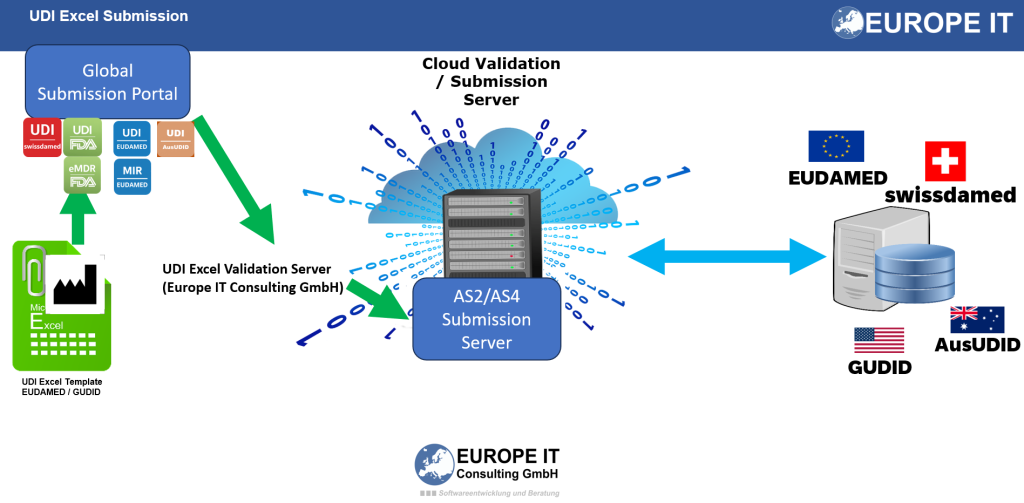
Comprehensive service offering for your AusUDID compliance
- AusUDID UDI Excel template: The professional basis for your structured UDI data entry for Australia.
- Data review and validation: We support you in the expert review of your UDI data so that typical inconsistencies can be identified and corrected at an early stage.
- Technical preparation: The structured Excel data can be used as a basis for technical conversion, upload processes or M2M preparation.
- AusUDID submission service: We accompany the process from data entry and review through to preparation and support of the AusUDID submission.
Technical specifications of the Excel template
- File format: Microsoft Excel (.xlsx)
- Compatibility: Microsoft Excel 2016 and later
- Structure: One main tab, five functional additional tabs and several code lists
- Data validation: Drop-downs and controlled value lists for consistent entries
- Key logic: Linking of the additional tabs via the Primary Device Identifier
- Updates: Adaptation of the template in the event of relevant changes to data requirements or technical specifications
Please complete the form to request an offer
Why leading medical technology companies trust our solution
Europe IT Consulting GmbH is your partner for regulatory IT solutions, UDI data management and technical data submissions. We support companies in preparing complex UDI data in a structured way and making it efficiently usable for regulatory databases.
What makes our AusUDID Excel template unique:
- Precise structuring of AusUDID-relevant data elements
- Clear separation between the main data record and functional additional information
- Controlled code lists for standardised entries
- Reduction of validation errors through predefined field logic
- Optimal preparation for manual review, technical conversion and M2M processes
- Practical usability for Regulatory Affairs, Quality Management, IT and external partners
- ANALYSE: Identify which AusUDID data are relevant for your products.
- STRUCTURE: Record your UDI data in a clear tab logic.
- VALIDATE: Check mandatory fields, conditional fields and controlled selectable values.
- SUBMIT: Use the structured data as the basis for technical preparation, upload or M2M processes.
Perfectly aligned with your individual requirements
For small and medium-sized product portfolios:
The Excel template provides a clear basis for structured data entry and later review.
For large UDI portfolios:
The separated tab structure helps to prepare even extensive product data volumes cleanly and to maintain additional product-related information.
For companies with existing FDA GUDID or EUDAMED data:
Existing UDI data sets can be used as a starting point. However, the data must still be reviewed from a subject-matter perspective, adapted to the Australian requirements and actively submitted to AusUDID.
For M2M-oriented processes:
Structured data entry forms an important basis for technical conversion, HL7 SPL / XML preparation and further automated submission processes.
Frequently asked questions to support your decision
Can I use existing UDI data from other markets?
Yes, existing UDI data from FDA GUDID, EUDAMED or internal UDI master data can serve as a basis. However, they must be checked and supplemented for the AusUDID requirements.
Can I use the Excel template to generate M2M files myself?
No, the Excel template primarily serves structured data entry. We offer technical conversion into a suitable exchange format as a separate service.
Why are there several tabs?
Not all information belongs in the main data record. Sterilisation methods, clinical sizes, storage conditions, package identifiers and documents can occur multiple times or conditionally. The separated tabs enable a clean and scalable data structure.
What role does the Primary Device Identifier play?
The Primary Device Identifier is the central key. It is used to clearly assign the information from the additional tabs to the correct product.
What are the CL tabs?
CL tabs are code lists. They contain controlled values for drop-down fields, e.g. risk classes, issuing agencies, sterilisation methods, MRI safety status or units of measure.
What happens if a column is added or deleted?
The structure should not be changed. Additional or deleted columns may impair technical further processing and the upload process.
Who should complete the template?
Typically, Regulatory Affairs, Quality Management, Master Data Management, IT and, where applicable, the Australian sponsor work together. Many fields require subject-matter coordination between manufacturer and sponsor.
Act now – prepare your AusUDID data early!
The Australian UDI requirements are becoming mandatory step by step. Companies that structure, validate and technically prepare their UDI data at an early stage reduce later correction loops, upload risks and operational bottlenecks.
Your first step towards efficient AusUDID compliance:
Please complete the form to request an offer
Invest now in a structured regulatory data strategy for Australia.
The AusUDID UDI Excel template is developed and distributed by Europe IT Consulting GmbH, your certified partner for regulatory IT solutions since 2011.
Note: Before starting the project, please review the current TGA requirements, deadlines, roles, data requirements and technical specifications. Existing UDI data from other markets can serve as a starting point, but do not replace subject-matter review and active submission to AusUDID.