The long-term benefits of UDI

The process of UDI implementation according to EUDAMED and/or FDA requirements is time-consuming and costly. However, there are significant benefits that go far beyond mere compliance. As a manufacturer and/or…

The process of UDI implementation according to EUDAMED and/or FDA requirements is time-consuming and costly. However, there are significant benefits that go far beyond mere compliance. As a manufacturer and/or…
The format of your UDI codes, Basic UDI-DI and UDI-DI, depends on the selected Issuing Agency. For the two most commonly used Issuing Agencies, GS1 and HIBC, we have summarized…

On the 8th February 2021 the European Commission published a Guidance on the Management of the Legacy Devices. It explains in detail how Legacy Devices will be identified and the…
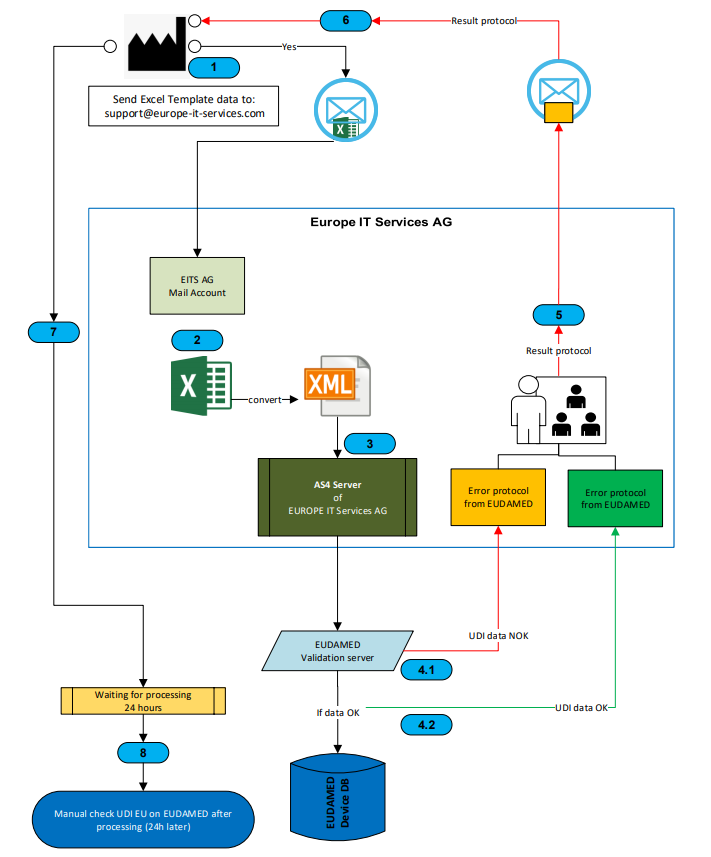
By using the Europe IT submission method, you can easily submit your UDI data to EUDAMED via Excel. But before starting to consider the data transfer process, make sure you…

Many economic operators have questions in the course of their UDI implementation, which are also asked by other economic operators. We have summarized and answered the most frequently asked questions…

Compared to other European countries, many German economic operators have not yet been assigned their Single Registration Number (SRN), and there are reasons for this. The Federal Ministry of Health…

You may have noticed that the UDI rules are quite complex and involve different actors. If you are in the MedTech industry, it is even more important to be clear…

Handlungsbedarf für Medizinproduktehersteller im Bereich UDI GUDID / EUDAMED Was für Lebensmittel heute bereits gilt, wird auch für Medizinprodukte Realität. Die Rückverfolgbarkeit muss sichergestellt werden, mit dem übergeordneten Ziel, die…
https://www.youtube.com/watch?v=5-3xiXmfjhg&feature=youtu.beLearn in this short video how our global UDI software solution helps you ensure a smooth and successful UDI implementation.