Single Use Device
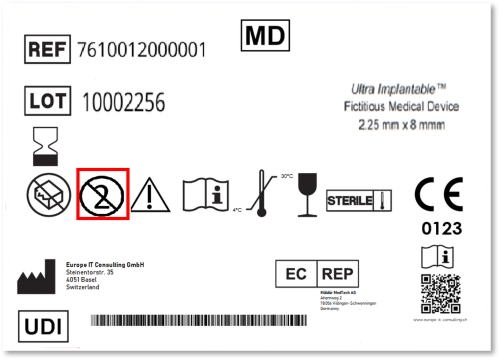
Since the EU Medical Device Regulation (MDR) came into force, the Unique Device Identification (UDI) system has become a cornerstone of regulatory compliance. For manufacturers of Single Use Devices (SUDs),…

Since the EU Medical Device Regulation (MDR) came into force, the Unique Device Identification (UDI) system has become a cornerstone of regulatory compliance. For manufacturers of Single Use Devices (SUDs),…

The EU has designated several UDI issuing entities (Issuing Entities) to provide manufacturers with various options for generating and managing their Unique Device Identifiers (UDIs). The currently recognized entities are:…

What is a "New Device" according to MDR and IVDR? Introduction The Medical Device Regulation (MDR, EU 2017/745) and the In Vitro Diagnostic Regulation (IVDR, EU 2017/746) have significantly tightened…

What is an "Active Device" according to MDR and IVDR? Introduction The Medical Device Regulation (MDR, EU 2017/745) and In Vitro Diagnostic Regulation (IVDR, EU 2017/746) have tightened the requirements…
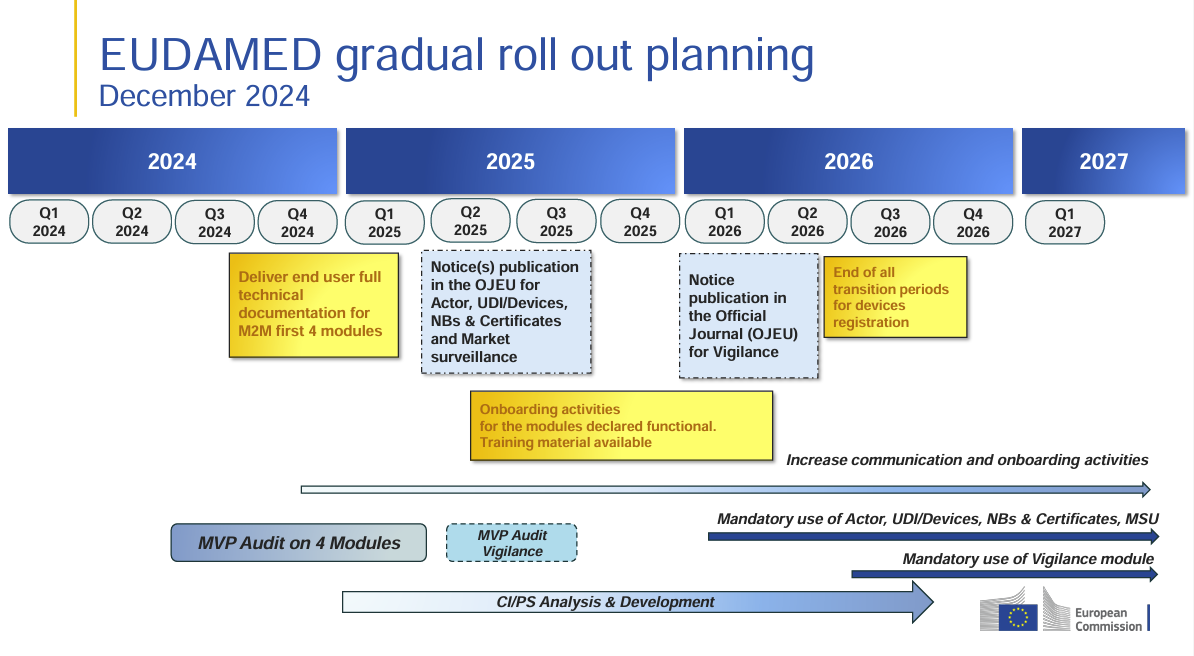
EUDAMED: Updated Rollout Plan for 2025-2027 Released The European Commission has released an updated EUDAMED rollout plan, introducing changes to the implementation timeline and transition periods. Companies in the MedTech…

Revolution or Hype? The MedTech industry faces the challenge of balancing regulatory requirements, efficient production processes, and innovative digital transformation. SAP serves as the backbone of IT infrastructure for many…

Latest Updates from Australia’s TGA The Australian Therapeutic Goods Administration (TGA) has made significant progress in implementing the Unique Device Identification (UDI) system for medical devices. We are pleased to…

FDA Updates GUDID Guidelines: Transition to GMDN Codes Updated FDA Guidelines for the Global Unique Device Identification Database (GUDID) The U.S. Food and Drug Administration (FDA) has released a new…

Dear Customers, Partners, and Friends, During this special time of the year, we would like to extend our warmest Christmas greetings to you. Thank you for your trust and support…

In 2025, numerous exciting events in the field of medical technology and regulatory affairs await us. For companies and professionals in the industry, these trade fairs and congresses provide…