
EUDAMED deadline 28 May 2026:
When a new UDI-DI brings forward the registration of the Basic UDI-DI
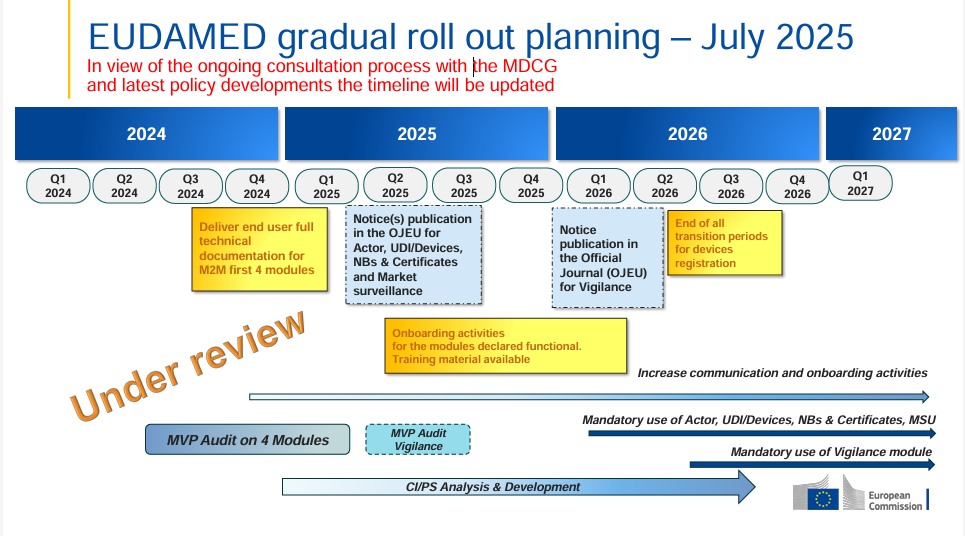
In just a few weeks, on 28 May 2026, the use of the first four EUDAMED modules will become mandatory. These include, among others, the Actor Registration Module and the UDI/Devices Registration Module. With the publication of Commission Decision (EU) 2025/2371, the European Commission confirmed the functionality of these modules; this results in mandatory use from 28 May 2026.
Many manufacturers have based their planning on the additional transitional period until 27 November 2026 for products already placed on the market – and this is generally understandable. The European Commission’s EUDAMED Q&A clarifies that, for products whose first unit with a specific UDI-DI was already placed on the EU market before the date of mandatory use of the UDI/Devices module, the corresponding registration must be completed within twelve months from the publication of the notice confirming functionality.
However, there is one specific scenario that is still not sufficiently considered in many Regulatory Affairs departments and may, in the worst case, block the marketability of a new product variant.
The scenario
An existing product family was lawfully placed on the market before the deadline and is assigned to a Basic UDI-DI. For the registration of the existing products or existing UDI-DIs, there is generally still time until November 2026.
Now, after 28 May 2026, a new variant is to be introduced – in other words, a new UDI-DI under the same existing Basic UDI-DI.
What applies in this case?
The clear regulatory position
There is no additional transitional period for this new UDI-DI.
The European Commission’s EUDAMED Q&A specifies the timelines at the level of the specific UDI-DI. If the first unit of a Regulation Device with a specific UDI-DI is placed on the EU market on or after the date of mandatory use of the UDI/Devices module, the corresponding registration in the UDI/DEV module must be completed before the first placing on the market.
This means that a newly added UDI-DI cannot automatically rely on the transitional period of the existing product family. What matters is not only that the product family already exists, but when the specific UDI-DI is placed on the market for the first time.
The transitional period until November 2026 applies to products or UDI-DIs whose first unit was already lawfully placed on the EU market before the deadline and for which further units of the same UDI-DI continue to be placed on the market after the deadline.
The technical consequence: The Basic UDI-DI must be available
This is where the real practical risk lies.
EUDAMED follows a clear parent-child structure: The Basic UDI-DI forms the parent level, under which one or more UDI-DIs are managed. The European Commission explicitly describes this structure: One or more UDI-DIs may be linked to a Basic UDI-DI; additional UDI-DIs may be added at a later stage. At the same time, each UDI-DI inherits attributes from the linked Basic UDI-DI.
In practice, this means that anyone who needs to register a new UDI-DI after 28 May 2026 also needs the corresponding Basic UDI-DI to be available in the system.
In this case, the Basic UDI-DI must be registered earlier from a technical and procedural perspective, even if older UDI-DIs within the same product family could theoretically still fall under the transitional period until November 2026.
This technical logic is also supported by the EUDAMED Business Rules: For a UDI-DI, exactly one Basic UDI-DI must exist, and several UDI-DIs may be assigned to the same Basic UDI-DI. In addition, a new UDI-DI can only be added to an existing Basic UDI-DI if the Basic UDI-DI has the status “Submitted” or “Registered”.
Particular risk with XML bulk upload and M2M transmission
Manufacturers transmitting UDI data via XML bulk upload or through an M2M interface should review this point at an early stage.
If a data record references a UDI-DI to a Basic UDI-DI that does not yet exist in EUDAMED, has not been correctly created, or is not yet in the required status, the transmission may fail during validation. The root cause is not always immediately apparent during error analysis – especially when several Basic UDI-DIs, many UDI-DIs or complex packaging structures are processed at the same time.
This can lead to unnecessary delays, particularly for larger product families, many variants or automated transmission processes.
Our practical recommendation
If a product launch with a new UDI-DI is planned after 28 May 2026, this new variant should not be considered in isolation.
A structured review of the entire product family is recommended:
- Which Basic UDI-DI is affected?
- Which existing UDI-DIs are already assigned to this Basic UDI-DI?
- Which new UDI-DI is to be introduced after the deadline?
- Is the Basic UDI-DI already available in EUDAMED?
- Does the Basic UDI-DI have the required status?
- Has the technical transmission sequence for XML or M2M been correctly prepared?
In many cases, it is more efficient to prepare and transmit the complete product family as one closed and validated data set. This reduces fragmentation, helps avoid referencing errors and creates regulatory clarity for later changes or additional variants.
Note on the source basis
This specific constellation – “a new UDI-DI under an existing Basic UDI-DI after the EUDAMED deadline” – is not described as a separate special case in the official documents.
However, the assessment results directly from the combination of three official sources:
First, from the European Commission’s EUDAMED Q&A, according to which the registration timeline is considered at the level of the specific UDI-DI. If the first unit with a specific UDI-DI is placed on the market on or after the date of mandatory use of the UDI/Devices module, the registration must be completed before the first placing on the market.
Second, from the EUDAMED structure between Basic UDI-DI and UDI-DI, according to which one or more UDI-DIs are linked to a Basic UDI-DI and each UDI-DI inherits attributes from the linked Basic UDI-DI.
Third, from the EUDAMED Business Rules, according to which a UDI-DI must be assigned to exactly one Basic UDI-DI and a new UDI-DI can only be added to an existing Basic UDI-DI if the Basic UDI-DI has the status “Submitted” or “Registered”.
Conclusion
The EUDAMED transitional periods should not only be considered at the level of the product family. The decisive factor is when the specific UDI-DI is placed on the market for the first time.
For new UDI-DIs from 28 May 2026 onwards, the rule is clear: Registration in the EUDAMED UDI/Devices module must be completed before the first placing on the market.
This also makes the corresponding Basic UDI-DI technically relevant. It must be available and correctly created in EUDAMED for the registration of the new UDI-DI – even if older UDI-DIs within the same product family may still fall under the transitional period until November 2026.
Europe IT Consulting supports manufacturers in the structured preparation, validation and transmission of UDI data to EUDAMED – both via Excel/XML-based processes and through M2M interfaces.
If you would like to check whether your current data records and upload structures are prepared for this scenario, please contact us.
Sources
- European Commission: Publication on the mandatory use of the first four EUDAMED modules from 28 May 2026.
https://health.ec.europa.eu/latest-updates/eudamed-four-first-modules-will-be-mandatory-use-28-may-2026-2025-11-27_en - European Commission: EUDAMED Overview – mandatory use of the first four modules, including Actor Registration and UDI/Device Registration.
https://health.ec.europa.eu/medical-devices-eudamed/overview_en - European Commission: UDI/Device Registration – submission of UDI/device information in EUDAMED.
https://health.ec.europa.eu/medical-devices-eudamed/udidevice-registration_en - European Commission: Q&A on the gradual roll-out of EUDAMED – timelines for the UDI/DEV module at UDI-DI level.
https://health.ec.europa.eu/document/download/0e7327c7-0e06-4fbd-90d3-8ab7bb30fe9f_en?filename=md_eudamed-qa_en.pdf - European Commission: EUDAMED UDI Concept – structure of Basic UDI-DI, UDI-DI and Package UDI-DI.
https://health.ec.europa.eu/system/files/2021-11/md_eudamed-udi-concept_en_0.pdf - European Commission: EUDAMED UDI/Device Business Rules – assignment of UDI-DI to Basic UDI-DI and addition of new UDI-DIs.
https://health.ec.europa.eu/system/files/2022-08/md_udi-device-business-rules_en_0.pdf








Related Posts